 | |
| Names | |
|---|---|
| Preferred IUPAC name
Phenanthridine[1] | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.005.396 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C13H9N | |
| Molar mass | 179.217 g/mol |
| Melting point | 107.4 °C (225.3 °F; 380.5 K) |
| Boiling point | 348.9 °C (660.0 °F; 622.0 K) |
| slightly soluble[2] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
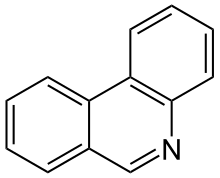
Phenanthridine is a nitrogen heterocyclic compound that is the basis of DNA-binding fluorescent dyes through intercalation. Examples of such dyes are ethidium bromide and propidium iodide. It is an isomer of acridine.
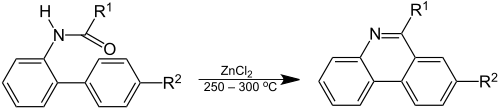
Phenanthridine was discovered by Amé Pictet and H. J. Ankersmit in 1891 by pyrolysis of the condensation product of benzaldehyde and aniline.[3] In the Pictet–Hubert reaction (1899) the compound is formed in a reaction of the 2-aminobiphenyl – formaldehyde adduct (an N-acyl-o-xenylamine) with zinc chloride at elevated temperatures.[4]
The reaction conditions for the Pictet–Hubert reaction were improved by Morgan and Walls in 1931, replacing the metal by phosphorus oxychloride and using nitrobenzene as a reaction solvent.[5] For this reason, the reaction is also called the Morgan–Walls reaction.[6]
The reaction is similar to the Bischler–Napieralski reaction and the Pictet–Spengler reaction.
References
- ↑ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. p. 212. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ↑ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, FL: CRC Press, pp. 3–460, ISBN 0-8493-0594-2
- ↑ Mittheilung Ueber das Phenanthridin Amé Pictet, H. J. Ankersmit Chemisches Laboratorium der Universität Genf Justus Liebigs Annalen der Chemie Volume 266 Issue 1–2, pp. 138–153 doi:10.1002/jlac.18912660107
- ↑ Mittheilungen Ueber eine neue Synthese der Phenanthridinbasen Amé Pictet, A. Hubert Berichte der deutschen chemischen Gesellschaft Volume 29 Issue 2, pp. 1182–1189, 1896 doi:10.1002/cber.18960290206
- ↑ CCCXXXV.—Researches in the phenanthridine series. Part I. A new synthesis of phenanthridine homologues and derivatives Gilbert T. Morgan, Leslie Percy Walls, J. Chem. Soc., 1931, 2447–2456 doi:10.1039/JR9310002447
- ↑ Jie Jack Li (ed.), 2004, Name Reactions in Heterocyclic Chemistry, Wiley.