| Soil-transmitted helminthiasis | |
|---|---|
| Other names | STH |
 | |
| Adult ascaris worms being removed from the bile duct of a patient in South Africa | |
Soil-transmitted helminthiasis is a type of worm infection (helminthiasis) caused by different species of roundworms. It is caused specifically by those worms which are transmitted through soil contaminated with faecal matter and are therefore called soil-transmitted helminths. Three types of soil-transmitted helminthiasis can be distinguished: ascariasis, hookworm infection and whipworm infection. These three types of infection are therefore caused by the large roundworm A. lumbricoides, the hookworms Necator americanus or Ancylostoma duodenale and by the whipworm Trichuris trichiura.
It has become the most common parasitic disease of humans worldwide.[1] Approximately two billion people (about a third of global population) are infected as of the latest estimate, and four billion at risk, surpassing even the all-time most prevalent parasitic disease, malaria.[2] The largest numbers of cases occur in impoverished rural areas of Sub-Saharan Africa, Latin America, Southeast Asia, and China.[3] Its main cause, like for many types of helminth infections, is lack of sanitation, such as the practice of open defecation, lack of hygiene such as hand washing and walking barefoot on contaminated soil.[4][5][6] It is regarded as one of the world's most important causes of intellectual and physical retardation.[7]
The helminthic disease is so named because the infection is transmitted through ingestion of the nematode eggs in the soil, which is contaminated through excrements. Therefore, the disease is most prevalent in warm and moist climates where sanitation and hygiene are poor and waters are unsafe, including the temperate zones during warmer months. STH is categorised among neglected tropical diseases because it inflicts tremendous disability and suffering, which can be clinically treated and relatively easily be prevented (primarily through improved sanitation), yet negligible attention has been given for many years.[8] It is now among the target diseases of London Declaration on Neglected Tropical Diseases (launched on 30 January 2012) to be controlled/eradicated by 2020.[9]
Simple prevention and control strategies are access to improved sanitation, public awareness on personal hygiene and health education.
Types
Soil-transmitted helminths are essentially intestinal parasites and their eggs which are liberated along with the faeces of infected persons into the soil. Ascaris and hookworm eggs become infective as they develop into larvae in soil. Infection occurs when vegetables and fruits, contaminated with soil infested eggs, are consumed; or when hands or fingers have been contaminated with dirt carrying the eggs are put in the mouth. On the other hand, hookworm eggs are not directly infective. They hatch in soil, releasing mobile larvae that can penetrate the skin. Thus infection is acquired through accidental contact with contaminated soil.[7]
Ascariasis
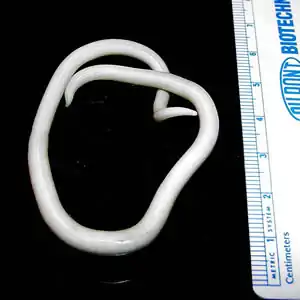
Ascariasis of STH is caused by the large roundworm A. lumbricoides. It is estimated to be the most widespread STH, affecting approximately 1 billion people. The victims constitute about half of the populations in tropical and subtropical areas. Most conditions are mild and often show little or no symptoms. Heavy infections however are debilitating, causing severe intestinal blockage and impair growth in children. Children, compounded with malnutrition, are most infected, with the most common age group being 3- to 8-year-olds, with an annual death of about 20,000. Children are more susceptible due to their frequent exposure to contaminated environment such as during playing, eating raw vegetables and fruits, and drinking wastewater.[7]
Hookworm disease
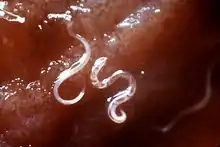
Hookworm infection of STH is caused by N. americanus and A. duodenale. Mild infections produce diarrhoea and abdominal pain. More severe infections can create serious health problems for newborns, children, pregnant women, and malnourished adults. In fact it is the leading cause of anaemia and protein deficiency in developing nations, affected an estimated 740 million people. N. americanus is the more common hookworm, while A. duodenale is more geographically restricted. Unlike other STHs, in which school-age children are most affected, high-intensity hookworm infections are more frequent in adults, specifically women. Roughly 44 million pregnant women are estimated to be infected. The disease causes severe adverse effects in both the mother and infant, such as low birth weight, impaired milk production, and increased risk of mortality.[7]
Trichuriasis

Whipworm (Trichuris trichiura) is the third most common STH-causing nematode in humans. According to current estimate, nearly 800 million people are infected, the majority of them children. Heavy infections could lead to acute symptoms such as diarrhoea and anaemia, and chronic symptoms such as growth retardation and impaired cognitive development. Medical conditions are more often serious since coinfection with protozoan parasites such as Giardia and Entamoeba histolytica, and with other nematodes is common.[7] Predominantly a tropical disease of developing countries, trichuriasis is quite common in the United States.[10]
Signs and symptoms
Symptoms becomes evident only when the intensity of infection is relatively high. Thus the degree of negative outcomes is directly related to worm burden; more worms means greater severity of disease.[11][12][13]
General
Most conditions of STH have a light worm burden and usually have no discernible symptoms. Heavy infections however cause a range of health problems, including abdominal pain, diarrhoea, blood and protein loss, rectal prolapse, and physical and mental retardation. Severe ascariasis is typically a pneumonia, as the larvae invade lungs, producing fever, cough and dyspnoea during early stage of infection.[14][15] Hookworm infections insinuate a skin reaction (dermatitis), increased white blood cells (eosinophils), a pulmonary reaction (pneumonitis), and skin rash (urticarial).[15][16]
Iron deficiency anaemia due to blood loss is a common symptom.[17]
Malnutrition
STH is often associated with malnutrition in children as it worsens their nutritional status in multiple ways.[2] The worms can induce intestinal bleeding, competition for nutrients (malabsorption of nutrients), frequent anaemia and diarrhoea.[18] Soil-transmitted helminths can also cause loss of appetite.[2] These nutritional "knock on" effects of STH can have a significant impact on the mental and physical development of children. In endemic countries, communities remain suppressed due to malnourishment, cognitive disability and physical weaknesses as a result of heavy infections.[11][12]
Diagnosis
For basic diagnosis, specific helminths can be generally identified from the faeces, and their eggs microscopically examined and enumerated using fecal egg count method. However, there are certain limitations such as the inability to identify mixed infections, and on clinical practice, the technique is inaccurate and unreliable.[19][20] A novel effective method for egg analysis is the Kato–Katz technique. It is a highly accurate and rapid method for A. lumbricoides and T. trichiura; however not so much for hookworm, which could be due to fast degeneration of the rather delicate hookworm eggs.[21]
Prevention
Prevention and control measures to prevent soil-transmitted helminthiasis are the following: availability of clean water for personal and domestic uses, improved access to sanitation which includes the use of properly functioning and clean toilets by all community members, education on personal hygiene such as hand washing and hygienic and safe food preparation; eliminating the use of untreated human faeces as fertilizer.[2]
Treatment
The World Health Organizations recommended albendazole or mebendazole for treatment.[2]
Mass treatment with drugs
One strategy to control the disease in areas where it is common is the treatment of entire groups of people regardless of symptoms via mass drug administration. The World Health Organization recommends mass treatments to all at-risk groups in endemic communities, especially women of childbearing age, and children. Mass treatments can also be provided to pregnant women in their second and third trimesters, and breastfeeding women.[22] This known as mass deworming.[2][23] While infected treating children is effective, there is significant evidence that concludes that routine deworming, in the absence of a positive test, does not improve nutrition, haemoglobin, school attendance or school performance.[24]
For this purpose, broad-spectrum benzimidazoles such as mebendazole and albendazole are the drugs of choice recommended by WHO. These anthelminthics are safe, inexpensive, and effective for several months. Mebendazole is given twice a day for three consecutive days, while albendazole is given as a single dose. WHO recommends annual treatment in areas where between 20 and 50% of people are infected, and a twice a year treatment if it is over 50%; and in low risk situations (i.e. less than 20% prevalence) case-by-case treatment.[17][25] In addition to these, pyrantel pamoate is also equally effective on ascaris. However, it has been reported that albendazole, mebendazole, and pyrantel pamoate are not entirely effective against T. trichiura with single oral doses in population-based control.[26]
Drugs for those with other diseases
In cases of coinfection, combination therapy with ivermectin and diethylcarbamazine is advocated. However coinfection with malaria and HIV, especially among African women, does not respond well to the current combination therapies.[27] It is more pressing for trichuriasis that the recommended drugs fail to provide positive results.[28] A novel drug tribendimidine, which was approved in China by the CCDC for human use in 2004, has been subjected to clinical trials showing that they are highly effective against major human flukes, ascaris (>90% cure rate) and hookworm (>82%); however with low cure rate for whipworm (<37%).[29]
Surgical intervention
In some severe cases of ascariasis the numbers of Ascaris worms can cause intestinal obstruction requiring emergency surgery.[30] The obstruction may be due to a collected mass of worms or to a twisting of the intestine.[30] During the surgery the worms may be manually removed.[30]
Epidemiology
Regions
Infections are widely distributed in tropical and subtropical areas, with the greatest numbers occurring in sub-Saharan Africa, the Americas, China and east Asia.[2]
Infection estimates
The World Health Organization estimates that globally more than 1.5 billion people (24% of the total population) have a soil-transmitted helminth infection.[2] Over 270 million preschool-age children and over 600 million school-age children live in areas where these parasites are intensively transmitted, and are in need of treatment and preventive interventions. Latest estimates indicate that more than 880 million children are in need of treatment from STH infections.[3][18][31]
By type of parasitic worm the breakdown is:[32]
Deaths
Latest estimates indicate that the total annual death toll which is directly attributable is as high as 135,000.[3][18][31] The death toll due to the malnutrition link is likely to be much higher.[33][34]
References
- ↑ Lo, Nathan C.; Heft-Neal, Sam; Coulibaly, Jean T.; Leonard, Leslie; Bendavid, Eran; Addiss, David G. (2019-11-01). "State of deworming coverage and equity in low-income and middle-income countries using household health surveys: a spatiotemporal cross-sectional study". The Lancet Global Health. 7 (11): e1511–e1520. doi:10.1016/S2214-109X(19)30413-9. ISSN 2214-109X. PMC 7024997. PMID 31558383.
- 1 2 3 4 5 6 7 8 "Soil-transmitted helminth infections Fact sheet N°366". who.int. April 2014. Retrieved 18 October 2014.
- 1 2 3 WHO (2012). Eliminating Soil-transmitted Helminthiasis as a Public Health Problem in Children: Progress Report 2001–2010 and Strategic Plan 2011–2020 (PDF). WHO Press, World Health Organization, Geneva, Switzerland. pp. 1–78. ISBN 978-92-4-150312-9.
- ↑ "CDC - Hookworm". 17 April 2019.
- ↑ Ziegelbauer K, Speich B, Mäusezahl D, Bos R, Keiser J, Utzinger J (January 2012). "Effect of sanitation on soil-transmitted helminth infection: systematic review and meta-analysis". PLOS Medicine. 9 (1): e1001162. doi:10.1371/journal.pmed.1001162. PMC 3265535. PMID 22291577.
- ↑ Strunz EC, Addiss DG, Stocks ME, Ogden S, Utzinger J, Freeman MC (March 2014). "Water, sanitation, hygiene, and soil-transmitted helminth infection: a systematic review and meta-analysis". PLOS Medicine. 11 (3): e1001620. doi:10.1371/journal.pmed.1001620. PMC 3965411. PMID 24667810.
- 1 2 3 4 5 Bethony J, Brooker S, Albonico M, Geiger SM, Loukas A, Diemert D, Hotez PJ (May 2006). "Soil-transmitted helminth infections: ascariasis, trichuriasis, and hookworm". Lancet. 367 (9521): 1521–32. doi:10.1016/S0140-6736(06)68653-4. PMID 16679166. S2CID 8425278.
- ↑ "Neglected Tropical Diseases". cdc.gov. June 6, 2011. Retrieved 28 November 2014.
- ↑ London Declaration (2012) (30 January 2012). "London Declaration on Neglected Tropical Diseases" (PDF). Retrieved 2013-03-26.
{{cite web}}: CS1 maint: numeric names: authors list (link) - ↑ Starr MC, Montgomery SP (October 2011). "Soil-transmitted Helminthiasis in the United States: a systematic review—1940–2010". The American Journal of Tropical Medicine and Hygiene. 85 (4): 680–4. doi:10.4269/ajtmh.2011.11-0214. PMC 3183777. PMID 21976572.
- 1 2 Hotez, Peter J.; Brindley, Paul J.; Bethony, Jeffrey M.; King, Charles H.; Pearce, Edward J.; Jacobson, Julie (2008). "Helminth infections: the great neglected tropical diseases". The Journal of Clinical Investigation. 118 (4): 1311–1321. doi:10.1172/JCI34261. PMC 2276811. PMID 18382743.
- 1 2 Mascarini-Serra, Luciene (2011). "Prevention of Soil-transmitted Helminth Infection". Journal of Global Infectious Diseases. 3 (2): 175–182. doi:10.4103/0974-777X.81696. PMC 3125032. PMID 21731306.
- ↑ Parija, Subhash Chandra; Chidambaram, Meenachi; Mandal, Jharna (2017). "Epidemiology and clinical features of soil-transmitted helminths". Tropical Parasitology. 7 (2): 81–85. doi:10.4103/tp.TP_27_17 (inactive 1 August 2023). PMC 5652059. PMID 29114484.
{{cite journal}}: CS1 maint: DOI inactive as of August 2023 (link) - ↑ Ramamoorthy, Karthik Ganesh (2015). "Anaesthesia and Ascaris pneumonia (Loeffler's syndrome)". Indian Journal of Anaesthesia. 59 (2): 125–126. doi:10.4103/0019-5049.151379. PMC 4357880. PMID 25788748.
- 1 2 Sarinas, P. S.; Chitkara, R. K. (1997). "Ascariasis and hookworm". Seminars in Respiratory Infections. 12 (2): 130–137. PMID 9195678.
- ↑ Loukas, Alex; Hotez, Peter J.; Diemert, David; Yazdanbakhsh, Maria; McCarthy, James S.; Correa-Oliveira, Rodrigo; Croese, John; Bethony, Jeffrey M. (2016). "Hookworm infection". Nature Reviews. 2: 16088. doi:10.1038/nrdp.2016.88. PMID 27929101.
- 1 2 WHO (2012). Helminth Control in School-age Children: a Guide for Managers of Control Programmes (PDF) (2 ed.). WHO Press, World Health Organization, Geneva, Switzerland. pp. 1–75. ISBN 978-92-4-154826-7.
- 1 2 3 Yap P, Fürst T, Müller I, Kriemler S, Utzinger J, Steinmann P (August 2012). "Determining soil-transmitted helminth infection status and physical fitness of school-aged children". Journal of Visualized Experiments. 66 (66): e3966. doi:10.3791/3966. PMC 3486755. PMID 22951972.
- ↑ Humphries D, Nguyen S, Boakye D, Wilson M, Cappello M (October 2012). "The promise and pitfalls of mass drug administration to control intestinal helminth infections". Current Opinion in Infectious Diseases. 25 (5): 584–9. doi:10.1097/QCO.0b013e328357e4cf. PMID 22903231. S2CID 34575855.
- ↑ Krauth SJ, Coulibaly JT, Knopp S, Traoré M, N'Goran EK, Utzinger J (2012). "An in-depth analysis of a piece of shit: distribution of Schistosoma mansoni and hookworm eggs in human stool". PLOS Neglected Tropical Diseases. 6 (12): e1969. doi:10.1371/journal.pntd.0001969. PMC 3527364. PMID 23285307.
- ↑ Tarafder MR, Carabin H, Joseph L, Balolong E, Olveda R, McGarvey ST (March 2010). "Estimating the sensitivity and specificity of Kato-Katz stool examination technique for detection of hookworms, Ascaris lumbricoides and Trichuris trichiura infections in humans in the absence of a 'gold standard'". International Journal for Parasitology. 40 (4): 399–404. doi:10.1016/j.ijpara.2009.09.003. PMC 2829363. PMID 19772859.
- ↑ "WHO | Strategy". WHO. Archived from the original on May 9, 2007. Retrieved 2019-10-03.
- ↑ Mascarini-Serra L (April 2011). "Prevention of Soil-transmitted Helminth Infection". Journal of Global Infectious Diseases. 3 (2): 175–82. doi:10.4103/0974-777X.81696. PMC 3125032. PMID 21731306.
- ↑ Taylor-Robinson, David C.; Maayan, Nicola; Donegan, Sarah; Chaplin, Marty; Garner, Paul (11 September 2019). "Public health deworming programmes for soil-transmitted helminths in children living in endemic areas". The Cochrane Database of Systematic Reviews. 9 (11): CD000371. doi:10.1002/14651858.CD000371.pub7. ISSN 1469-493X. PMC 6737502. PMID 31508807.
- ↑ WHO (2006). Preventive Chemotherapy in Human Helminthiasis : Coordinated Use of Anthelminthic Drugs in Control Interventions : a Manual for Health Professionals and Programme Managers (PDF). WHO Press, World Health Organization, Geneva, Switzerland. pp. 1–61. ISBN 978-9241547109.
- ↑ Keiser J, Utzinger J (April 2008). "Efficacy of current drugs against soil-transmitted helminth infections: systematic review and meta-analysis". JAMA. 299 (16): 1937–48. doi:10.1001/jama.299.16.1937. PMID 18430913.
- ↑ Ivan E, Crowther NJ, Rucogoza AT, Osuwat LO, Munyazesa E, Mutimura E, Njunwa KJ, Zambezi KJ, Grobusch MP (December 2012). "Malaria and helminthic co-infection among HIV-positive pregnant women: prevalence and effects of antiretroviral therapy". Acta Tropica. 124 (3): 179–84. doi:10.1016/j.actatropica.2012.08.004. PMID 22940013.
- ↑ Speich B, Ame SM, Ali SM, Alles R, Hattendorf J, Utzinger J, Albonico M, Keiser J (2012). "Efficacy and safety of nitazoxanide, albendazole, and nitazoxanide-albendazole against Trichuris trichiura infection: a randomized controlled trial". PLOS Neglected Tropical Diseases. 6 (6): e1685. doi:10.1371/journal.pntd.0001685. PMC 3367984. PMID 22679525.
- ↑ Xiao SH, Utzinger J, Tanner M, Keiser J, Xue J (May 2013). "Advances with the Chinese anthelminthic drug tribendimidine in clinical trials and laboratory investigations". Acta Tropica. 126 (2): 115–26. doi:10.1016/j.actatropica.2013.01.009. PMID 23352956.
- 1 2 3 Hefny AF, Saadeldin YA, Abu-Zidan FM (May 2009). "Management algorithm for intestinal obstruction due to ascariasis: a case report and review of the literature". Ulusal Travma ve Acil Cerrahi Dergisi = Turkish Journal of Trauma & Emergency Surgery. 15 (3): 301–5. PMID 19562557.
- 1 2 Lustigman S, Prichard RK, Gazzinelli A, Grant WN, Boatin BA, McCarthy JS, Basáñez MG (2012). "A research agenda for helminth diseases of humans: the problem of helminthiases". PLOS Neglected Tropical Diseases. 6 (4): e1582. doi:10.1371/journal.pntd.0001582. PMC 3335854. PMID 22545164.
- ↑ Hotez PJ, Fenwick A, Savioli L, Molyneux DH (May 2009). "Rescuing the bottom billion through control of neglected tropical diseases". Lancet. 373 (9674): 1570–5. doi:10.1016/S0140-6736(09)60233-6. PMID 19410718. S2CID 18371227.
- ↑ Sangalang, Stephanie O.; Medina, Shelley Anne J.; Ottong, Zheina J.; Lemence, Allen Lemuel G.; Totanes, Donrey; Valencia, John Cedrick; Singson, Patricia Andrea A.; Olaguera, Mikaela; Prado, Nelissa O.; Ocaña, Roezel Mari Z.; Canja, Rovin James F. (2020-12-30). "Protocol for a Trial Assessing the Impacts of School-Based WaSH Interventions on Children's Health Literacy, Handwashing, and Nutrition Status in Low- and Middle-Income Countries". International Journal of Environmental Research and Public Health. 18 (1): 226. doi:10.3390/ijerph18010226. PMC 7795080. PMID 33561075.
- ↑ Muslim, Azdayanti; Lim, Yvonne Ai-Lian; Mohd Sofian, Sakinah; Shaari, Syahrul Azlin; Mohd Zain, Zaini (2021). "Nutritional status, hemoglobin level and their associations with soil-transmitted helminth infections between Negritos (indigenous) from the inland jungle village and resettlement at town peripheries". PLOS ONE. 16 (1): e0245377. Bibcode:2021PLoSO..1645377M. doi:10.1371/journal.pone.0245377. PMC 7806132. PMID 33439889.