| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Triethylborane | |||
| Other names
Triethylborine, triethylboron | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider | |||
| ECHA InfoCard | 100.002.383 | ||
| EC Number |
| ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
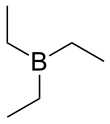
| (CH3CH2)3B | |||
| Molar mass | 98.00 g/mol | ||
| Appearance | Colorless liquid | ||
| Density | 0.677 g/cm3 | ||
| Melting point | −93 °C (−135 °F; 180 K) | ||
| Boiling point | 95 °C (203 °F; 368 K) | ||
| Not applicable; highly reactive | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards |
Spontaneously flammable in air; causes burns | ||
| GHS labelling: | |||
 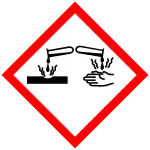   | |||
| Danger | |||
| H225, H250, H301, H314, H330, H360 | |||
| P201, P202, P210, P222, P233, P240, P241, P242, P243, P260, P264, P270, P271, P280, P281, P284, P301+P310, P301+P330+P331, P302+P334, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P310, P320, P321, P330, P363, P370+P378, P403+P233, P403+P235, P405, P422, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | < −20 °C (−4 °F; 253 K) | ||
| −20 °C (−4 °F; 253 K) | |||
| Safety data sheet (SDS) | External SDS | ||
| Related compounds | |||
Related compounds |
| ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |||
Triethylborane (TEB), also called triethylboron, is an organoborane (a compound with a B–C bond). It is a colorless pyrophoric liquid. Its chemical formula is (CH3CH2)3B or (C2H5)3B, abbreviated Et3B. It is soluble in organic solvents tetrahydrofuran and hexane.
Preparation and structure
Triethylborane is prepared by the reaction of trimethyl borate with triethylaluminium:[1]
- Et3Al + (MeO)3B → Et3B + (MeO)3Al
The molecule is monomeric, unlike H3B and Et3Al, which tend to dimerize. It has a planar BC3 core.[1]
Applications
Turbojet engine
Triethylborane was used to ignite the JP-7 fuel in the Pratt & Whitney J58 turbojet/ramjet engines powering the Lockheed SR-71 Blackbird[2] and its predecessor, the A-12 OXCART. Triethylborane is suitable because it ignites readily upon exposure to oxygen. It was chosen as an ignition method for reliability reasons, and in the case of the Blackbird, because JP-7 fuel has very low volatility and is difficult to ignite. Conventional ignition plugs posed a high risk of malfunction. Triethylborane was used to start each engine and to ignite the afterburners.[3]
Rocket
Mixed with 10–15% triethylaluminium, it was used before lift-off to ignite the F-1 engines on the Saturn V rocket.[4]
The Merlin engines that power the SpaceX Falcon 9 rocket use a triethylaluminium-triethylborane mixture (TEA-TEB) as a first- and second-stage ignitor.[5]
The Firefly Aerospace Alpha launch vehicle's Reaver engines are also ignited by a triethylaluminium-triethylborane mixture.[6]
Organic chemistry
Industrially, triethylborane is used as an initiator in radical reactions, where it is effective even at low temperatures.[1] As an initiator, it can replace some organotin compounds.
It reacts with metal enolates, yielding enoxytriethylborates that can be alkylated at the α-carbon atom of the ketone more selectively than in its absence. For example, the enolate from treating cyclohexanone with potassium hydride produces 2-allylcyclohexanone in 90% yield when triethylborane is present. Without it, the product mixture contains 43% of the mono-allylated product, 31% di-allylated cyclohexanones, and 28% unreacted starting material.[7] The choice of base and temperature influences whether the more or less stable enolate is produced, allowing control over the position of substituents. Starting from 2-methylcyclohexanone, reacting with potassium hydride and triethylborane in THF at room temperature leads to the more substituted (and more stable) enolate, whilst reaction at −78 °C with potassium hexamethyldisilazide, KN[Si(CH
3)
3]
2 and triethylborane generates the less substituted (and less stable) enolate. After reaction with methyl iodide the former mixture gives 2,2-dimethylcyclohexanone in 90% yield while the latter produces 2,6-dimethylcyclohexanone in 93% yield.[7][8] The Et stands for ethyl group CH3CH2−.

It is used in the Barton–McCombie deoxygenation reaction for deoxygenation of alcohols. In combination with lithium tri-tert-butoxyaluminum hydride it cleaves ethers. For example, THF is converted, after hydrolysis, to 1-butanol. It also promotes certain variants of the Reformatskii reaction.[9]
Triethylborane is the precursor to the reducing agents lithium triethylborohydride ("Superhydride") and sodium triethylborohydride.[10]
- MH + Et3B → MBHEt3 (M = Li, Na)
Triethylborane reacts with methanol to form diethyl(methoxy)borane, which is used as the chelating agent in the Narasaka–Prasad reduction for the stereoselective generation of syn-1,3-diols from β-hydroxyketones.[11][12]
Safety
Triethylborane is strongly pyrophoric, with an autoignition temperature of −20 °C (−4 °F),[13] burning with an apple-green flame characteristic for boron compounds. Thus, it is typically handled and stored using air-free techniques. Triethylborane is also acutely toxic if swallowed, with an LD50 of 235 mg/kg in rat test subjects.[14]
See also
References
- 1 2 3 Brotherton, Robert J.; Weber, C. Joseph; Guibert, Clarence R.; Little, John L. (15 June 2000). "Boron Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Wiley-VCH. doi:10.1002/14356007.a04_309. ISBN 3527306730.
- ↑ "Lockheed SR-71 Blackbird". March Field Air Museum. Archived from the original on 2000-03-04. Retrieved 2009-05-05.
- ↑ "Lockheed SR-71 Blackbird Flight Manual". www.sr-71.org. Archived from the original on 2011-02-02. Retrieved 2011-01-26.
- ↑ A. Young (2008). The Saturn V F-1 Engine: Powering Apollo Into History. Springer. p. 86. ISBN 978-0-387-09629-2.
- ↑ Mission Status Center, June 2, 2010, 1905 GMT Archived May 30, 2010, at the Wayback Machine, SpaceflightNow, accessed 2010-06-02, Quotation: "The flanges will link the rocket with ground storage tanks containing liquid oxygen, kerosene fuel, helium, gaseous nitrogen and the first stage ignitor source called triethylaluminum-triethylborane, better known as TEA-TEB."
- ↑ "https://twitter.com/Firefly_Space/status/1090319933534334977". Twitter. Retrieved 2023-02-05.
{{cite web}}: External link in|title= - 1 2 Crich, David, ed. (2008). "Enoxytriethylborates and Enoxydiethylboranes". Reagents for Radical and Radical Ion Chemistry. Handbook of Reagents for Organic Synthesis. Vol. 11. John Wiley & Sons. ISBN 9780470065365. Archived from the original on 2022-02-19. Retrieved 2019-01-27.
- ↑ Negishi, Ei-ichi; Chatterjee, Sugata (1983). "Highly regioselective generation of "thermodynamic" enolates and their direct characterization by NMR". Tetrahedron Letters. 24 (13): 1341–1344. doi:10.1016/S0040-4039(00)81651-2.
- ↑ Yamamoto, Yoshinori; Yoshimitsu, Takehiko; Wood, John L.; Schacherer, Laura Nicole (15 March 2007). "Triethylborane". Encyclopedia of Reagents for Organic Synthesis. Wiley. doi:10.1002/047084289X.rt219.pub3. ISBN 978-0471936237.
- ↑ Binger, P.; Köster, R. (1974). "Sodium triethylhydroborate, sodium tetraethylborate, and sodium triethyl-1-propynylborate". Inorganic Syntheses. 15: 136–141. doi:10.1002/9780470132463.ch31. ISBN 9780470132463.
- ↑ Chen, Kau-Ming; Gunderson, Karl G.; Hardtmann, Goetz E.; Prasad, Kapa; Repic, Oljan; Shapiro, Michael J. (1987). "A Novel Method for the In situ Generation of Alkoxydialkylboranes and Their Use in the Selective Preparation of 1,3-syn Diols". Chemistry Letters. 16 (10): 1923–1926. doi:10.1246/cl.1987.1923.
- ↑ Yang, Jaemoon (2008). "Diastereoselective Syn-Reduction of β-Hydroxy Ketones". Six-Membered Transition States in Organic Synthesis. John Wiley & Sons. pp. 151–155. ISBN 9780470199046. Archived from the original on 2022-02-19. Retrieved 2019-01-27.
- ↑ "Fuels and Chemicals - Autoignition Temperatures". Archived from the original on 2015-05-04. Retrieved 2017-08-26.
- ↑ "Archived copy". Archived from the original on 2022-02-19. Retrieved 2020-09-26.
{{cite web}}: CS1 maint: archived copy as title (link)