 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
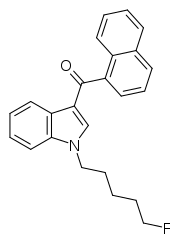
| Formula | C24H22FNO |
| Molar mass | 359.444 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
AM-2201 (1-(5-fluoropentyl)-3-(1-naphthoyl)indole) is a recreational designer drug that acts as a potent but nonselective full agonist for the cannabinoid receptor.[2] It is part of the AM series of cannabinoids discovered by Alexandros Makriyannis at Northeastern University.
Hazards
Convulsions have been reported[3] including at doses as low as 10 mg.[4]
Pharmacology
AM-2201 is a full agonist for cannabinoid receptors. Affinities are: with a Ki of 1.0 nM at CB1 and 2.6 nM at CB2.[5] The 4-methyl functional analog MAM-2201 probably has similar affinities. AM-2201 has an EC50 of 38 nM for human CB1 receptors, and 58 nM for human CB2 receptors.[6] AM-2201 produces bradycardia and hypothermia in rats at doses of 0.3–3 mg/kg, comparable to the potency of JWH-018 in rats, suggesting potent cannabinoid-like activity.[6]
Pharmacokinetics
AM-2201 metabolism differs only slightly from that of JWH-018. AM-2201 N-dealkylation produces fluoropentane instead of pentane (or plain alkanes in general).
Detection
A forensic standard of AM-2201 is available, and the compound has been posted on the Forendex website of potential drugs of abuse.[7]
Legal status
In the United States, AM-2201 is a Schedule I controlled substance.[8]
See also
References
- ↑ Anvisa (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.
- ↑ Wilkinson SM, Banister, Kassiou M (2015). "Bioisosteric Fluorine in the Clandestine Design of Synthetic Cannabinoids". Australian Journal of Chemistry. 68 (1): 4–8. doi:10.1071/CH14198.
- ↑ McQuade D, Hudson S, Dargan PI, Wood DM (March 2013). "First European case of convulsions related to analytically confirmed use of the synthetic cannabinoid receptor agonist AM-2201". European Journal of Clinical Pharmacology. 69 (3): 373–6. doi:10.1007/s00228-012-1379-2. PMID 22936123. S2CID 23136932.
- ↑ ekaJ (20 February 2011). "The Night I Killed My Friends". Erowid.org. Retrieved 11 June 2012.
- ↑ WO patent 0128557, Makriyannis A, Deng H, "Cannabimimetic indole derivatives", granted 2001-06-07
- 1 2 Banister SD, Stuart J, Kevin RC, Edington A, Longworth M, Wilkinson SM, Beinat C, Buchanan AS, Hibbs DE, Glass M, Connor M, McGregor IS, Kassiou M (August 2015). "Effects of bioisosteric fluorine in synthetic cannabinoid designer drugs JWH-018, AM-2201, UR-144, XLR-11, PB-22, 5F-PB-22, APICA, and STS-135". ACS Chemical Neuroscience. 6 (8): 1445–58. doi:10.1021/acschemneuro.5b00107. PMID 25921407.
- ↑ "Southern Association of Forensic Scientists". Archived from the original on 2014-09-10. Retrieved 2013-07-16.
- ↑ Controlled Substances listed by the DEA