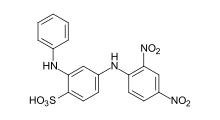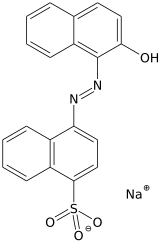
An acid dye is a dye that is typically applied to a textile at low pH. They are mainly used to dye wool, not cotton fabrics.[1] Some acid dyes are used as food colorants,[2][3] and some can also be used to stain organelles in the medical field.
Acid dyes are anionic, soluble in water and are essentially applied from acidic bath. These dyes possess acidic groups, such as SO3H and COOH and are applied on wool, silk and nylon when ionic bond is established between protonated –NH2 group of fibre and acid group of dye. Overall wash fastness is poor although lightfastness is quite good. As dye and fibre contain opposite electrical nature, strike rate and uptake of acid dye on these fibres is faster; electrolyte at higher concentration is added to retard dye uptake and to form levelled shades. Acid generates cation on fibre and temperature helps to substitute negative part of acid with anionic dye molecules.[4]
Description
Acid dyes are generally divided into three classes according to their fastness requirements, migration ability, and dyeing pH.[5]
Acid dyes affix to fibers by hydrogen bonding, Van der Waals forces[6] and ionic bonding. While some acid dyes work in water, many choose to activate dyes in acid dye-baths instead. According to the Brønsted–Lowry acid–base theory, an acid is a molecule or ion capable of donating a proton, and this is determined by the acid dissociation constant. Compared to most acids, water has a much higher pKa value, meaning that it dissociates to give H+ with more difficulty. In this context, if an acid is used instead of water, then the hydrogen ion (H+) is more easily able to dissociate in order to react with the aniline dye anion, allowing the dye to dissolve.
Animal protein fibers and the synthetic fiber nylon contain many cationic sites that bind anionic dye. The strength (fastness) of this bond reflects the strength of this ionic interaction.
Uses
Fibers
In the laboratory, home, or art studio, the acid used in the dye-bath is often vinegar (acetic acid) or citric acid. The uptake rate of the dye is controlled with the use of sodium chloride. In textiles, acid dyes are effective on protein fibers, i.e. animal hair fibers like wool, alpaca, and mohair. They are also effective on silk.[7] They are effective in dyeing the synthetic fiber nylon, but of minimum interest in dyeing any other synthetic fibers.
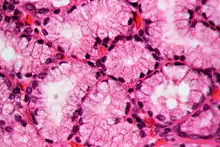

Histology
In staining during microscopic examination for diagnosis or research, acid dyes are used to color basic tissue proteins. In contrast, basic dyes are used to stain cell nuclei and some other acidic components of tissues.[8] Regarding cellular structures, acid dyes will stain acidophilic structures that have a net positive charge due to the fact that they have a negatively charged chromophore. Acidophilic structures include the cytoplasm, collagen and mitochondria. The two have an affinity for each other due to the conflicting charges.[9][10] Examples of acid dyes used in medicine include:[11]
- Lee's stain (stains reddish-pink).
- Phosphotungstic Acid Hematoxylin (PTAH) stain (stains blue).
- Eosin stain (stains pinkish-orange).
Food Industry
Acid dyes can also be used as food colouring, helping to increase the attractiveness of certain foods, and thus becoming more appealing to customers. Some examples include erythrosine, tartrazine, sunset yellow and allura red, to name a few, many of which are azo dyes.[12] These dyes can be used in frosting, cookies, bread, condiments or drinks. In order to prevent health hazards, a dye must be approved for consumption before it can be marked as edible. Some separation methods that can be used to identify unapproved dyes include the solid phase extraction process, the overpressured thin layer chromatography process, and the use of reversed-phase plates.[13]
Structures
The chemistry of acid dyes is complex and diverse. Most acid dyes are related in basic structure to the following:
- Anthraquinone type: Many acid dyes are synthesized from chemical intermediates that form anthraquinone-like structures as their final state. Many blue dyes have this structure as their basic shape. The structure predominates in the leveling class of acid dye.
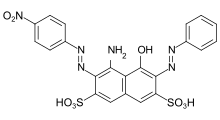
- Azo dyes: The structure of azo dyes contains the azo group (R-N=N−R. Most azo dyes are not acid dyes, but many acid dyes are azo dyes. Many acid dyes of the azo type are red in color.[14]
- Triarylmethane dye: These predominate in the milling class of dye. There are many yellow and green dyes commercially applied to fibers that are related to triphenylmethane.
Classes of acid dyes
Acidic dyes can be classified according to their dyeing behavior. This includes their wet fastness, migration ability, and dyeing pH:[1]
- Leveling acid dyes: These dyes have relatively low molecular weights. Consequently, they migrate more readily before fixation and they exhibit low wet fastness. They are not normally suited for use as apparel fabric. They require an acidic dye bath, often using sulfuric acid and sodium sulfate mixtures (pH2-4),[7] together with leveling agents such as ethoxylated fatty amines.
- Milling dyes: These dyes are high molecule weight, with the result that migrates slowly. Consequently, they exhibit wet fastness, which is useful for dyeing wool materials. Milling acid dyes are sometimes called 'Neutral acid dyes' as they do not require an acidic dye bath. They are commonly applied using Acetic acid (pH4-7).[7]
- Metal complex acid dyes: These dyes are composed of acid dye molecules complexed with a metal ion, which will usually be chromium or cobalt. Metal complex acid dyes have high molecular weights, giving them low mobility and high wet fastness. Due to this, they are commonly used on nylon and synthetic polyamide fibers. Metal complex acid dyes are economical. However, they produce relatively dull shades. Metal complex acid dyes take a larger range of pH in the dyebath (pH2-7).[7]
Safety
Some dyes are mutagenic and carcinogenic, including methyl orange, acid red 26, and trypan blue.[15][16]
References
- 1 2 Booth, Gerald (2000). "Dyes, General Survey". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a09_073. ISBN 3527306730.
- ↑ Trowbridge Filippone, Peggy. "Food Color Additives". Archived from the original on April 5, 2015. Retrieved September 8, 2016.
- ↑ Klaus Hunger, ed. (2003), Industrial Dyes: Chemistry, Properties, Applications (in German), Weinheim: WILEY-VCH Verlag, pp. 276ff, ISBN 978-3-662-01950-4
- ↑ A K Roy Choudhary, “Textile Preparation and Dyeing”, Science Publishers, USA (2006)
- ↑ "Mechanism of Dyeing with Acid Dyes". Textile Learner. Mazharul Islam Kiron. Retrieved 2012-01-08.
- ↑ Clark, Jim (2012). "Intermolecular bonding - van der Waals forces". chemguide.co.uk. Retrieved 15 June 2014.
- 1 2 3 4 "How Acid Dye Works". Retrieved October 21, 2019.
- ↑ Bruckner, Monica Z. "Basic Cellular Staining". Retrieved December 12, 2013.
- ↑ "Staining and Commonly Used Stains". Histology Learning system. Boston University. Retrieved 2019-11-05.
- ↑ Gokhale, S (2008). Pharmaceutical Biology. Maharashtra, India: Pragati Books Pvt. Ltd.
- ↑ "Staining and Commonly Used Stains". Histology Learning system. Boston University. Retrieved 2019-11-05.
- ↑ Frazier, R.A (2007). CAPILLARY ELECTROPHORESIS | Food Additives. Elsevier Ltd.
- ↑ Vega, M (2000). Encyclopedia of Separation Science. Elsevier Ltd.
- ↑ Hunger, Klaus; Mischke, Peter; Rieper, Wolfgang; Raue, Roderich; Kunde, Klaus; Engel, Aloys (2005). "Azo Dyes". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_245. ISBN 3527306730.
- ↑ Prival, M. J.; Bell, S. J.; Mitchell, V. D.; Peiperl, M. D.; Vaughan, V. L. (1984). "Mutagenicity of benzidine and benzidine-congener dyes and selected monoazo dyes in a modified Salmonella assay". Mutation Research. 136 (1): 33–47. doi:10.1016/0165-1218(84)90132-0. PMID 6371512.
- ↑ Bansal, Megha; Yadav, Rajesh Kumar (2016). "OCCUPATIONAL HEALTH HAZARDS AND AWARENESS OF OCCUPATIONAL SAFETY AMONG WORKERS OF TEXTILE DYEING INDUSTRIES IN JAIPUR, INDIA". Suresh Gyan Vihar University International Journal of Environment, Science and Technology. 2 (2): 30–38. S2CID 37596329.
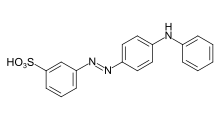
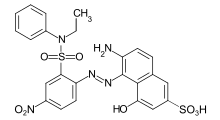
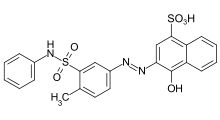
.svg.png.webp)