 | |
 | |
| Names | |
|---|---|
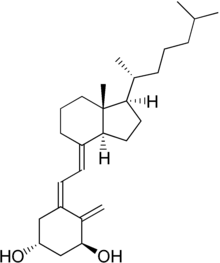
| Preferred IUPAC name
(1R,3S,5Z)-4-Methylidene-5-[(2E)-2-{(1R,3aS,7aR)-7a-methyl-1-[(2R)-6-methylheptan-2-yl]octahydro-4H-inden-4-ylidene}ethylidene]cyclohexane-1,3-diol | |
| Other names
Alphacalcidol; 1-Hydroxycholecalciferol | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.050.253 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C27H44O2 | |
| Molar mass | 400.64 g/mol |
| Melting point | 136 °C (277 °F; 409 K) |
| 0.016 g/100 mL | |
| Pharmacology | |
| A11CC03 (WHO) | |
| License data | |
| Legal status | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Alfacalcidol (or 1-hydroxycholecalciferol) is an analogue of vitamin D used for supplementation in humans and as a poultry feed additive.
Alfacalcidol has a weaker impact on calcium metabolism[3] and parathyroid hormone levels[4] than calcitriol; but significant effects on the immune system, including regulatory T cells.[5] It is considered to be a more useful form of vitamin D supplementation, mostly due to much longer half-life and lower kidney load.[6] It is the most commonly prescribed vitamin D metabolite for patients with end stage renal disease, given that impaired renal function alters the ability to carry out the second hydroxylation step required for the formation of the physiologically active form of vitamin D, 1,25-dihydroxyvitamin D3. Alfacalcidol is an active vitamin D3 metabolite, and therefore does not require the second hydroxylation step in the kidney.[7]
It was patented in 1971 and approved for medical use in 1978.[8]
Trade names
Pharmaceutical trade names include AlphaD and One-Alpha.
Other animals
Used as a poultry feed additive, it prevents tibial dyschondroplasia and increases phytate bioavailability.[9]
References
- ↑ "One-Alpha Capsules - Summary of Product Characteristics (SmPC)". (emc). 9 November 2017. Retrieved 17 February 2021.
- ↑ "List of nationally authorised medicinal products" (PDF). ema.europa.eu. 11 February 2021. Retrieved 14 April 2023.
- ↑ Cavalli L, Cavalli T, Marcucci G, Falchetti A, Masi L, Brandi ML (May 2009). "Biological effects of various regimes of 25-hydroxyvitamin D3 (calcidiol) administration on bone mineral metabolism in postmenopausal women". Clinical Cases in Mineral and Bone Metabolism. 6 (2): 169–73. PMC 2781231. PMID 22461169.
- ↑ Moe S, Wazny LD, Martin JE (2008). "Oral calcitriol versus oral alfacalcidol for the treatment of secondary hyperparathyroidism in patients receiving hemodialysis: a randomized, crossover trial". The Canadian Journal of Clinical Pharmacology. 15 (1): e36-43. PMID 18192704. Archived from the original on 2014-10-06. Retrieved 2013-12-04.
- ↑ Zold E, Szodoray P, Nakken B, Barath S, Kappelmayer J, Csathy L, et al. (January 2011). "Alfacalcidol treatment restores derailed immune-regulation in patients with undifferentiated connective tissue disease". Autoimmunity Reviews. 10 (3): 155–62. doi:10.1016/j.autrev.2010.09.018. hdl:2437/180066. PMID 20868777.
- ↑ Nuti R, Bianchi G, Brandi ML, Caudarella R, D'Erasmo E, Fiore C, et al. (March 2006). "Superiority of alfacalcidol compared to vitamin D plus calcium in lumbar bone mineral density in postmenopausal osteoporosis". Rheumatology International. 26 (5): 445–53. doi:10.1007/s00296-005-0073-4. PMID 16283320. S2CID 9931256.
- ↑ Ritzerfeld M, Klasser M, Mann H (December 2001). "Alfacalcidol in the therapy of renal bone disease". International Journal of Clinical Pharmacology and Therapeutics. 39 (12): 546–50. doi:10.5414/cpp39546. PMID 11770836.
- ↑ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. XXX. ISBN 9783527607495.
- ↑ "Vitamin Derivatives". vitaminderivatives.com. Retrieved 14 April 2023.