 | |
| Names | |
|---|---|
| IUPAC name
N5-(1-Hydroxycyclopropyl)-L-glutamine | |
| Systematic IUPAC name
(2S)-2-Amino-5-[(1-hydroxycyclopropyl)amino]-5-oxopentanoic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H14N2O4 | |
| Molar mass | 202.210 g·mol−1 |
| Melting point | 197 to 199 °C (387 to 390 °F; 470 to 472 K)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Coprine is a mycotoxin. It was first isolated from common inkcap (Coprinopsis atramentaria). It occurs in mushrooms in the genera Coprinopsis.[2] When combined with alcohol, it causes "Coprinus syndrome".[3]: 284 [4] It inhibits the enzyme acetaldehyde dehydrogenase, which is involved in the metabolism of alcohol. This inhibition leads to a buildup of acetaldehyde, causing an alcohol flush reaction. Because of this, the mushroom is commonly referred to as Tippler's Bane.
History
Because of the similarities to disulfram (tetraethylthiuram disulfide) poisoning, it was long speculated that disulfram was the active ingredient in common inkcap. In 1956 it was reported that disulfram had been isolated from coprinus, but this finding could not be replicated.[5] In 1975, coprine was identified as the compound in the common inkcap, with the mechanism identified in 1979.[6][7][8]
Symptoms
Symptoms of coprine poisoning include facial reddening/flushing, nausea, vomiting, malaise, agitation, palpitations, tingling in limbs, and sometimes headache and excessive salivation. [3]: 288 [9] This can be described as the alcohol flush reaction. Symptoms typically arise five to ten minutes after consumption of alcohol. If no more alcohol is consumed, the symptoms will generally subside over two to three hours, and symptom severity is proportional to the amount of alcohol consumed. Consumption of alcohol can induce these symptoms for up to 5 days after ingesting coprine.[9] Interestingly, symptoms of coprine poisoning do not appear when the mushroom is ingested raw, but only when the mushroom is cooked.[5]
In examining coprine poisoning cases in Germany in 2010, none of the patients died, and all made full recoveries after abstaining from alcohol. In one case medical care was not sought at all, and while there was a range in time of ethanol consumption after mushroom consumption, all the cases had well-cooked the mushrooms before ingestion.[10]
The symptoms of coprine poisoning and alcohol consumption are similar to those induced by disulfiram (marketed as Antabuse), a drug utilized to treat chronic alcoholism by inducing severe side-effects to alcohol consumption. Because of this, research was done into the use of coprine as a similar drug for alcoholism. [11] However, testing has shown coprine to have long-term mutagenic and reproductive effects, making it ill-suited for long-term use. [12]
Mechanism of action
Coprine hydrolyzes to glutamic acid and 1-aminocyclopropanol, which inhibits the enzyme acetaldehyde dehydrogenase.[11] 1-aminocyclopropanol quickly converts to cyclopropanone hydrate, which binds covalently to the thiol group present in the enzyme, deactivating the dehydrogenase activity.[8] This inhibition then causes a buildup of acetaldehyde if ethanol is ingested. Since acetaldehyde is toxic and can no longer be metabolized to the less toxic acetic acid, the characteristic symptoms of coprine poisoning occur. However, as shown in the mechanism below, the covalent bonding is reversible, which is what allows symptoms to subside if no more alcohol is consumed.[8]
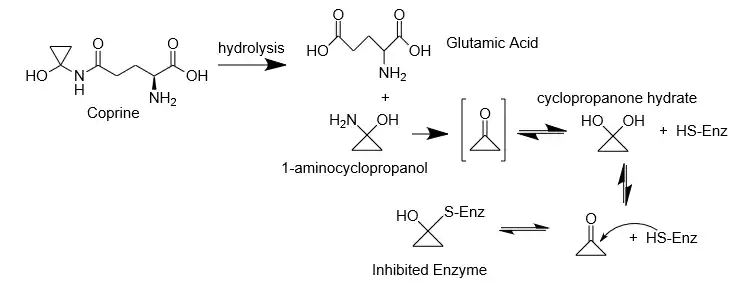
1-Aminocyclopropanol also deactivates the esterase activity of acetaldehyde dehydrogenase, but less significantly.[13]
Synthesis
Coprine is the first discovered compound with a naturally occurring cyclopropanone group.[7] Chemical synthesis can be effectively carried out by conducting an N-acylation reaction on 1-aminocyclopropanol.[7] Treatment of isocyanatocyclopropane with hydrochloric acid leads to the hydrochloride of 1-aminocyclopropanol. Adding sodium hydroxide to create 1-aminocyclopropanol will destabilize the structure, so synthesis must be conducted using the hydrochloride. The addition of the hydrochloride to N-phthaloyl-L-glutamic anhydride will undergo acylation. Lastly, the blocking group is removed using hydrazine, yielding coprine.[7] The enantiomer, isocoprine, is formed in negligible quantities in small-scale synthesis but is synthesized in higher amounts in large-scale, industrial synthesis.[7]
References
- ↑ RÖMPP Online – Version 3.4, Stuttgart: Thieme Chemistry, 2009
- ↑ "Disulfiramlike Mushroom Toxicity". Medscape. 2017-01-07.
- 1 2 Benjamin, Denis R. (1995). Mushrooms: poisons and panaceas—a handbook for naturalists, mycologists and physicians. New York: WH Freeman and Company. ISBN 978-0-7167-2600-5.
- ↑ Michelot, D. (1992). "Poisoning by Coprinus atramentarius". Natural Toxins. 1 (2): 73–80. doi:10.1002/nt.2620010203. PMID 1344910.
- 1 2 Buck, Robert W. (5 October 1961). "Mushroom Toxins — A Brief Review of the Literature". New England Journal of Medicine. 265 (14): 681–686. doi:10.1056/NEJM196110052651406. PMC 1574797.
- ↑ Lindberg, Per; Bergman, Rolf; Wickberg, Börje (1975). "Isolation and structure of coprine, a novel physiologically active cyclopropanone derivative from Coprinus atramentarius and its synthesis via 1-aminocyclopropanol". Journal of the Chemical Society, Chemical Communications (23): 946. doi:10.1039/C39750000946.
- 1 2 3 4 5 Lindberg, Per; Bergman, Rolf; Wickberg, Börje (1977). "Isolation and structure of coprine, the in vivo aldehyde dehydrogenase inhibitor in Coprinus atramentarius; syntheses of coprine and related cyclopropanone derivatives". Journal of the Chemical Society, Perkin Transactions 1 (6): 684. doi:10.1039/P19770000684.
- 1 2 3 Wiseman, Jeffrey S.; Abeles, Robert H. (May 2002). "Mechanism of inhibition of aldehyde dehydrogenase by cyclopropanone hydrate and the mushroom toxin coprine". Biochemistry. 18 (3): 427–435. doi:10.1021/bi00570a006. PMID 369602.
- 1 2 "Mushroom Poisoning Syndromes". North American Mycological Association. Retrieved 23 April 2020.
- ↑ Haberl, Bettina; Pfab, Rudolf; Berndt, Sigmar; Greifenhagen, Christoph; Zilker, Thomas (3 March 2011). "Case series: Alcohol intolerance with Coprine-like syndrome after consumption of the mushroom (Pers.:Fr.) Quél., 1886 (Freckled Dapperling)". Clinical Toxicology. 49 (2): 113–114. doi:10.3109/15563650.2011.554840. PMID 21370948. S2CID 43434106.
- 1 2 Koppaka, Vindhya; Thompson, David C.; Chen, Ying; Ellermann, Manuel; Nicolaou, Kyriacos C.; Juvonen, Risto O.; Petersen, Dennis; Deitrich, Richard A.; Hurley, Thomas D.; Vasiliou, Vasilis; Sibley, David R. (July 2012). "Aldehyde Dehydrogenase Inhibitors: a Comprehensive Review of the Pharmacology, Mechanism of Action, Substrate Specificity, and Clinical Application". Pharmacological Reviews. 64 (3): 520–539. doi:10.1124/pr.111.005538. PMC 3400832. PMID 22544865.
- ↑ Michelot, Didier (1992). "Poisoning byCoprinus atramentarius". Natural Toxins. 1 (2): 73–80. doi:10.1002/nt.2620010203. PMID 1344910.
- ↑ Marchner, Hans; Tottmar, Olof (July 1983). "Studies in vitro on the inactivation of mitochondrial rat-liver aldehyde dehydrogenase by the alcohol-sensitizing compounds cyanamide, 1-aminocyclopropanol and disulfiram". Biochemical Pharmacology. 32 (14): 2181–2188. doi:10.1016/0006-2952(83)90224-1. PMID 6870943.