 | |
 | |
| Names | |
|---|---|
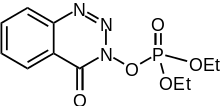
| Preferred IUPAC name
Diethyl 4-oxo-1,2,3-benzotriazin-3(4H)-yl phosphate | |
| Other names
DEPBT | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.156.337 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C11H14N3O5P | |
| Molar mass | 299.22 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
DEPBT (3-(diethoxyphosphoryloxy)-1,2,3-benzotriazin-4(3H)-one) is a peptide coupling reagent used in peptide synthesis. It shows remarkable resistance to racemization.[1]
Fmoc-Dab(Mtt)-OH, a commercially available amino acid building block for solid-phase peptide synthesis (SPPS), was proven to undergo rapid lactamization, instead of reacting with the N-terminal end of the peptide. Compared with other commercially available coupling reagents, DEPBT has shown superior performance in coupling Fmoc-Dab(Mtt)-OH to the N-terminal end of peptide during SPPS, though the approach was regarded as 'costly and tedious'.[2]
See also
References
- ↑ Li, Haitao; Jiang, Xiaohui; Ye, Yun-hua; Fan, Chongxu; Romoff, Todd; Goodman, Murray (1999). "3-(Diethoxyphosphoryloxy)-1,2,3- benzotriazin-4(3H)-one (DEPBT): A New Coupling Reagent with Remarkable Resistance to Racemization". Organic Letters. 1 (1): 91–94. doi:10.1021/ol990573k. ISSN 1523-7060. PMID 10822541.
- ↑ Lam, Pak-Lun; Wu, Yue; Wong, Ka-Leung (2022). "Incorporation of Fmoc-Dab(Mtt)-OH during solid-phase peptide synthesis: a word of caution". Organic & Biomolecular Chemistry. 20 (13): 2601–2604. doi:10.1039/D2OB00070A.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.