 | |
| Names | |
|---|---|
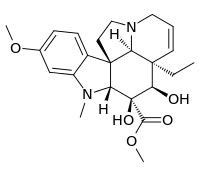
| IUPAC name
Methyl 3β,4β-dihydroxy-16-methoxy-1-methyl-6,7-didehydro-2β,5α,12β,19α-aspidospermidine-3-carboxylate | |
| Systematic IUPAC name
Methyl (3aR,3a1R,4R,5S,5aR,10bR)-3b-ethyl-4,5-dihydroxy-8-methoxy-6-methyl-3a,3a1,4,5,5a,6,11,12-octahydro-1H-indolizino[8,1-cd]carbazole-5-carboxylate | |
| Other names
Desacetylvindoline; 17-O-Deacetylvindoline | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C23H30N2O5 | |
| Molar mass | 414.502 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Deacetylvindoline is a terpene indole alkaloid produced by Catharanthus roseus. Deacetylvindoline is the product of a hydroxylation of desacetoxyvindoline by deacetoxyvindoline 4-hydroxylase (D4H).[1] It is a substrate for deacetylvindoline O-acetyltransferase (DAT) which acetylates a hydroxy group to form vindoline, one of the two immediate precursors for the formation of the pharmacetucially valuable bisindole alkaloid vinblastine.[2]
References
- ↑ Vazquez-Flota, De Carolis, Alarco and De Luca (1997) Molecular cloning and characterization of desacetoxyvindoline-4-hydroxylase, a 2-oxoglutarate dependent-dioxygenase involved in the biosynthesis of vindoline in Catharanthus roseus (L.) G. Don. Plant Molecular Biology. 34(6). 935-948
- ↑ Power, Kurz and De Luca (1990) Purification and characterization of acetylcoenzyme A: Deacetylvindoline 4-O-acetyltransferase from Catharanthus roseus. Archives of Biochemistry and Biophysics. 279(2). 370-376
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.