 | |
 | |
| Names | |
|---|---|
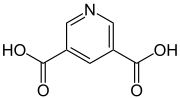
| Preferred IUPAC name
Pyridine-3,5-dicarboxylic acid | |
| Other names
3,5-Pyridinedicarboxylic acid | |
| Identifiers | |
3D model (JSmol) |
|
| 131640 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.007.177 |
| EC Number |
|
| 279307 | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C7H5NO4 | |
| Molar mass | 167.120 g·mol−1 |
| Structure[1] | |
| monoclinic | |
| P21/c, No. 14 | |
a = 9.702 Å, b = 11.153 Å, c = 6.587 Å α = 90°, β = 107.80°, γ = 90° | |
Formula units (Z) |
4 |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Dinicotinic acid (pyridine-3,5-dicarboxylic acid) is a heterocyclic organic compound, more precisely a heteroaromatic. It is one of many pyridinedicarboxylic acids and consists of a pyridine ring carrying to carboxy groups in the 3- and 5-positions.
Preparation and properties
Dinicotinic acid can be formed by heating pyridine-2,3,5,6-tetracarboxylic acid or carbodinicotinic acid (pyridine-2,3,5-tricarboxylic acid).[2][3]
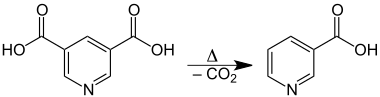
The acid is sparingly soluble in water and ether. Its melting point of 323 °C is the highest among pyridinedicarboxylic acids. Upon heating, it decarboxylates and decomposes to nicotinic acid:[4]
References
- ↑ Structure reports for 1973 : organic section. Organic compounds. J. Trotter. Dordrecht: Springer. 1975. p. 174. ISBN 978-94-017-3121-8. OCLC 859588799.
{{cite book}}: CS1 maint: others (link) - ↑ Meyer, Hans; Tropsch, Hans (1914). "Über Dinicotinsäure und deren Abbau zu ββ′-Diaminopyridin und über das αα′-Diaminopyridin". Monatshefte für Chemie (in German). 35 (2): 207–217. doi:10.1007/BF01518124. ISSN 0026-9247.
- ↑ Wolffenstein, Richard (1922). Die Pflanzenalkaloide (in German) (Dritte, verbesserte und vermehrte Auflage ed.). Berlin, Heidelberg. p. 67. ISBN 978-3-642-92449-1. OCLC 913710178.
{{cite book}}: CS1 maint: location missing publisher (link) - ↑ Alam, Mahbub; Khan, M. Hafeez (1980). "Preparation of some nicotinic acid derivatives". Philippine Journal of Science. 109 (1–2): 19–21.
See also
- Dipicolinic acid, an isomeric dicarboxylic acid
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.