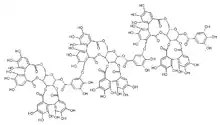
 | |
| Names | |
|---|---|
| Systematic IUPAC name
(10aR,11S,12aR,25aR,25bS)-2,3,4,5,6,7,17,18,19,20,21,22-Dodecahydroxy-9,15,24,27-tetraoxo-9,10a,11,12a,13,15,24,25a,25b,27-decahydrodibenzo[g,i]dibenzo[6′,7′:8′,9′][1,4]dioxecino[2′,3′:4,5]pyrano[3,2-b][1,5]dioxacycloundecin-11-yl (310aR,311S,312aR,325aR,325bS,810aR,811R,812aR,825aR,825bS)-15,16,32,33,34,35,36,37,317,318,319,320,321,322,64,65,82,83,84,85,86,87,817,818,819,820,821,822,113,114,115-hentriacontahydroxy-39,315,324,327,5,89,815,824,827,10-decaoxo-39,310a,311,312a,313,315,324,325a,325b,327,89,810a,811,812a,813,815,824,825a,825b,827-icosahydro-2,4,7,9-tetraoxa-3,8(22,11)-bis(dibenzo[g,i]dibenzo[6′,7′:8′,9′][1,4]dioxecino[2′,3′:4,5]pyrano[3,2-b][1,5]dioxacycloundecina)-1,11(1),6(1,3)-tribenzenaundecaphane-13-carboxylate | |
| Identifiers | |
3D model (JSmol) |
|
PubChem CID |
|
| |
| |
| Properties | |
| C123H80O78 | |
| Molar mass | 2805.915 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Lambertianin C is an ellagitannin.
Natural occurrence
Lambertianin C can be found in Rubus species such as Rubus lambertianus,[2] in cloudberries (Rubus chamaemorus)[3] and in red raspberries (Rubus idaeus).[4]
Chemistry
Lambertianin C is trimer of casuarictin linked by sanguisorbic acid ester groups between glucopyranose moieties.[2] It contributes to the in vitro antioxidant activity of raspberries.[5]
References
- ↑ "Lambertianin C". PubChem. 2017-07-29.
- 1 2 Tanaka, T.; Tachibana, H.; Nonaka, G.; Nishioka, I.; Hsu, F. L.; Kohda, H.; Tanaka, O. (1993). "Tannins and related compounds. CXXII. New dimeric, trimeric and tetrameric ellagitannins, lambertianins A-D, from Rubus lambertianus Seringe". Chemical and Pharmaceutical Bulletin. 41 (7): 1214–1220. doi:10.1248/cpb.41.1214. PMID 8374992.
- ↑ Kähkönen, M.; Kylli, P.; Ollilainen, V.; Salminen, J. P.; Heinonen, M. (2012). "Antioxidant Activity of Isolated Ellagitannins from Red Raspberries and Cloudberries". Journal of Agricultural and Food Chemistry. 60 (5): 1167–1174. doi:10.1021/jf203431g. PMID 22229937.
- ↑ Mullen, W.; Stewart, A. J.; Lean, M. E.; Gardner, P.; Duthie, G. G.; Crozier, A. (2002). "Effect of freezing and storage on the phenolics, ellagitannins, flavonoids, and antioxidant capacity of red raspberries". Journal of Agricultural and Food Chemistry. 50 (18): 5197–5201. doi:10.1021/jf020141f. PMID 12188629.
- ↑ Borges, G.; Degeneve, A.; Mullen, W.; Crozier, A. (2010). "Identification of Flavonoid and Phenolic Antioxidants in Black Currants, Blueberries, Raspberries, Red Currants, and Cranberries†". Journal of Agricultural and Food Chemistry. 58 (7): 3901–3909. doi:10.1021/jf902263n. PMID 20000747.
External links
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.