 | |
| Names | |
|---|---|
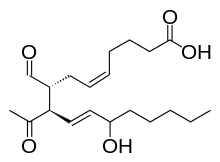
| IUPAC name
(5Z,8R,9R,10E,12S)-9-acetyl-8-formyl-12- hydroxyheptadeca-5,10-dienoic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| KEGG | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C20H32O5 | |
| Molar mass | 352.465 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
 | |
| Names | |
|---|---|
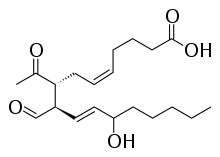
| IUPAC name
(5Z,8R,9R,10E,12S)-8-acetyl-9-formyl-12- hydroxyheptadeca-5,10-dienoic acid | |
| Other names
LGE2 | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| KEGG | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C20H32O5 | |
| Molar mass | 352.465 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Levuglandins are reactive aldehydes formed by the spontaneous rearrangement of prostaglandin H (PGH). Enantiomerically pure levuglandin (LG) E2 can also be formed through the cyclooxygenase (COX) pathway by a rearrangement of the prostaglandin (PG) endoperoxide PGH 2.[1] They are nonclassic eicosanoids. One species, levuglandin E2, (LGE2), forms neurotoxic adducts with amyloid beta.[2][3] Levuglandins and isolevuglandins can damage proteins by covalent adduction, thereby interfering with their normal functions. These lipid-derived protein modifications may serve as dosimeters of oxidative injury. Elevated plasma levels of isoLG-protein epitopes are associated with atherosclerosis but are independent of total cholesterol, a classical risk factor.
History
Though spontaneous rearrangements of PGH2 are known to generate prostaglandins (PG) PGD2 and PGE2.[4][5] Prof. Robert Salomon at Case Western Reserve University discovered that a novel alternative rearrangement also occurs that producing two γ-ketoaldehydes[6] and named them levuglandins LGD2 and LGE2 as they are derivatives of levulinaldehyde with prostanoid side chains.
References
- ↑ Salomon RG (2005). "Isolevuglandins, oxidatively truncated phospholipids, and atherosclerosis". Ann. N. Y. Acad. Sci. 1043: 327–42. Bibcode:2005NYASA1043..327S. doi:10.1196/annals.1333.040. PMID 16037255. S2CID 12782911.
- ↑ Boutaud, Olivier; Montine, Thomas J.; Chang, Lei; Klein, William L.; Oates, John A. (2006). "PGH2-derived levuglandin adducts increase the neurotoxicity of amyloid beta1-42". Journal of Neurochemistry. 96 (4): 917–923. doi:10.1111/j.1471-4159.2005.03586.x. PMC 1621054. PMID 16412101.
- ↑ Boutaud, O.; Brame, C. J.; Salomon, R. G.; Roberts Lj, 2nd; Oates, J. A. (1999). "Characterization of the lysyl adducts formed from prostaglandin H2 via the levuglandin pathway". Biochemistry. 38 (29): 9389–96. doi:10.1021/bi990470+. PMID 10413514.
{{cite journal}}: CS1 maint: numeric names: authors list (link) - ↑ Hamberg M., Samuelsson B. (1973). "Detection and isolation of an endoperoxide intermediate in prostaglandin biosynthesis". Proc. Natl. Acad. Sci. U.S.A. 70 (3): 899–903. Bibcode:1973PNAS...70..899H. doi:10.1073/pnas.70.3.899. PMC 433384. PMID 4514999.
- ↑ Nugteren D.H., Hazelhof E. (1973). "Isolation and properties of intermediates in prostaglandin biosynthesis". Biochim. Biophys. Acta. 326 (3): 448–461. doi:10.1016/0005-2760(73)90145-8. PMID 4776443.
- ↑ Salomon R.G.; Miller D.B.; Zagorski M.G.; Coughlin D.J. (1984). "Prostaglandin endoperoxides. 14. Solvent-induced fragmentation of prostaglandin endoperoxides. New aldehyde products from PGH2 and a novel intramolecular 1*2-hydride shift during endoperoxide fragmentation in aqueous solution". J. Am. Chem. Soc. 106 (20): 6049–6060. doi:10.1021/ja00332a049.