 | |
| Names | |
|---|---|
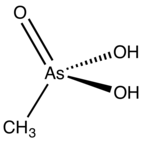
| Preferred IUPAC name
Methylarsonic acid[1] | |
| Other names
Methanearsonic acid Monomethylarsonic acid | |
| Identifiers | |
3D model (JSmol) |
|
| 4-04-00-03682 | |
| ChemSpider | |
| ECHA InfoCard | 100.004.278 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 1557 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| CH5AsO3 | |
| Molar mass | 139.970 g·mol−1 |
| Appearance | white solid |
| Melting point | 160.5 °C (320.9 °F; 433.6 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Methylarsonic acid is an organoarsenic compound with the formula CH3AsO3H2. It is a colorless, water-soluble solid. Salts of this compound, e.g. disodium methyl arsonate, have been widely used in as herbicides and fungicides in growing cotton and rice.[2]
Reactions
Near physiological pH, methanearsonic acid converts to its conjugate bases, the methylarsonates. These include CH3AsO3H− and CH
3AsO2−
3.
Synthesis and biosynthesis
Reaction of arsenous acid with methyl iodide gives methylarsonic acid. This historically significant conversion is called the Meyer reaction:[3]
- As(OH)3 + CH3I + NaOH → CH3AsO(OH)2 + NaI + H2O
The then-novel aspect of the reaction was that alkylation occurs at arsenic, leading to oxidation of arsenic from oxidation state +3 to +5.
The biomethylation of arsenic compounds is thought to start with the formation of methanearsonates. Thus, trivalent arsenic compounds are methylated to give methanearsonate. S-Adenosylmethionine is the methyl donor. The methanearsonates are the precursors to cacodylates, again by the cycle of reduction (to methylarsonous acid) followed by a second methylation.[4]
Safety
Like most arsenic compounds, it is highly toxic.[4]
References
- ↑ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. pp. 916, 918. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ↑ Grund, S. C.; Hanusch, K.; Wolf, H. U. "Arsenic and Arsenic Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_113.pub2. ISBN 978-3527306732.
- ↑ G. Meyer (1883). "Ueber einige anomale Reaktionen". Berichte der deutschen chemischen Gesellschaft. 13: 1439–1443. doi:10.1002/cber.188301601316.
- 1 2 Styblo, M.; Del Razo, L. M.; Vega, L.; Germolec, D. R.; LeCluyse, E. L.; Hamilton, G. A.; Reed, W.; Wang, C.; Cullen, W. R.; Thomas, D. J. (2000). "Comparative toxicity of trivalent and pentavalent inorganic and methylated arsenicals in rat and human cells". Archives of Toxicology. 74: 289–299. doi:10.1007/s002040000134.