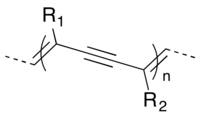
Polydiacetylenes (PDAs) are a family of conducting polymers closely related to polyacetylene. They are created by the 1,4 topochemical polymerization of diacetylenes. They have multiple applications from the development of organic films to immobilization of other molecules.[1]
History
The first polydiacetylene to be discovered was Poly(1,6-bishydroxy hexa-2,4-diacetylene) by Gerhard Wegner in 1969. This was achieved by exposing crystals of 1,6-bishydroxy hexa-2,4-diyne to UV light.[2] Polymerization was assumed to occur because of the spatial arrangement of diynes in the crystal, but this was not confirmed until 1972 when Raymond H. Baughman coined the term "topochemical polymerization" to describe polymerization due to spatial arrangement and put forth the spatial requirements needed for a polymerization of this sort.[3]
Synthesis

Synthesis of polydiacetylenes occurs through topochemical polymerization of 1,3-diynes. Typically, this must occur in the solid state, because many diynes undergo both 1,2 and 1,4 polymerization in solution – such is the case with diiodobutadiyne,[4] and other diynes with electron withdrawing substituents. The ideal arrangement of diynes in the solid state is a repeat distance of 5Å, a 45° tilt angle, and a 3.5Å distance between C1 of one monomer and C4 of the adjacent diyne monomer.[3]
Usually, polymerization is accompanied by a color change, due to the presence of an extended π-system. In addition, many PDAs exhibit thermochromism caused by twisting of the polymer backbone, changing the amount of conjugation in the system.[5] Depending upon the structure of the monomer, the resulting PDA can have interesting properties such as formation of a vesicle or tube structure.[6]
The chromatic transition from blue to red phase of PDA is caused by the electronic structure of PDAs' backbone which is featured by the alternative carbon double bond and carbon triple bond. Upon exposure to the external stimuli such as thermal, chemical and mechanical stimulus, the conjugation effect will endow the chromatic properties to this type material. As far as the pure or "intrinsic" PDAs are concerned, the blue to red phase transition is irreversible, however if the head groups of the PDAs' side chain are strengthened by other structure, for example chelation with metal oxide nanoparticles, it could provide resilience of the backbone to conduct the reversible color change.[7] Several recent researches which assisted by DFT simulation have shown that the chromatic properties could be modified by adjusting the side chain structures of PDA.[8] With the development thin film fabrication technology such as inkjet printing, PDAs could be coated on different substrate materials as multifunctional sensor, for example Kapton films, aluminum foil or even conventional paper.[9][10]
Zhu et al. have recently published an article about the synthesis of polydiacetylene using copper catalyst. This is first report on synthesis of polydiacetylenes in the solution phase.[11]
References
- ↑ Reppy, Mary A.; Pindzola, Bradford A. (2007). "Biosensing with polydiacetylene materials: structures, optical properties and applications". Chemical Communications (42): 4317–38. doi:10.1039/b703691d. PMID 17957278.
- ↑ Wegner, Gerhard (1969), "Topochemische Reaktionen von Monomeren mit konjugierten Dreifachbindungen / Tochemical Reactions of Monomers with conjugated triple Bonds", Zeitschrift für Naturforschung B, 24 (7): 824–832, doi:10.1515/znb-1969-0708, S2CID 98080997
- 1 2 Baughman, Raymond H. (1972), "Solid state polymerization of diacetylenes", Journal of Applied Physics, 43 (11): 4362–4378, Bibcode:1972JAP....43.4362B, doi:10.1063/1.1660929
- ↑ Luo, Liang; Wilhelm, Christopher; Sun, Aiwu; Grey, Clare P.; Lauher, Joseph W.; Goroff, Nancy S. (2008), "Poly(Diiododiacetylene): Preparation, Isolation, and Full Characterization of a Very Simple Poly(diacetylene)", Journal of the American Chemical Society, 130 (24): 7702–7709, doi:10.1021/ja8011403, PMID 18489101
- ↑ Xuemei Sun; Tao Chen; Sanqing Huang; Li Li; Huisheng Peng (2010). "Chromatic polydiacetylene with novel sensitivity". Chem. Soc. Rev. 39 (11): 4244–4257. doi:10.1039/C001151G. PMID 20877863. S2CID 5317564.
- ↑ Hsu, Te-Jung; Fowler, Frank W.; Lauher, Joseph (2012), "Preparation and Structure of a Tubular Addition Polymer: A True Synthetic Nanotube", Journal of the American Chemical Society, 134 (1): 142–145, doi:10.1021/ja209792f, PMID 22168532
- ↑ Wu, Aide; Beck, Christian; Federici, John; Iqbal, Zafar (3 September 2013). "Thermochromism in Polydiacetylene−ZnO Nanocomposites". J. Phys. Chem. C. 117 (38): 19593–19600. doi:10.1021/jp403939p.
- ↑ Wu, Aide; Gu, Yuan; Tian, Huiquan; Federici, John; Iqbal, Zafar (12 August 2014). "Effect of alkyl chain length on chemical sensing of polydiacetylene and polydiacetylene/ZnO nanocomposites". Colloid Polym Sci. 292 (12): 3137–3146. doi:10.1007/s00396-014-3365-y. S2CID 95537167.
- ↑ Wu, Aide; Gu, Yuan; Beck, Christian; Iqbal, Zafar; Federici, John (28 November 2013). "Reversible chromatic sensor fabricated by inkjet printing TCDA-ZnO on a paper substrate". Sens. Actuator B-Chem. 193: 10–18. doi:10.1016/j.snb.2013.10.130.
- ↑ Wu, Aide; Gu, Yuan; Starvou, Constantino; Kazerani, Hamed; Federici, John; Iqbal, Zafar (8 July 2014). "Inkjet printing colorimetric controllable and reversiblepoly-PCDA/ZnO composites". Sens. Actuator B-Chem. 203: 320–326. doi:10.1016/j.snb.2014.06.132.
- ↑ Sun, Han-Li; Wu, Bin; Liu, Da-Qi; Yu, Zi-Di; Wang, Jun-Jie; Liu, Qianyi; Liu, Xingchen; Niu, Dawen; Dou, Jin-Hu; Zhu, Rong (2022). "Synthesis of Polydiynes via an Unexpected Dimerization/Polymerization Sequence of C3 Propargylic Electrophiles". Journal of the American Chemical Society. 144 (19): 8807–8817. doi:10.1021/jacs.2c02816.