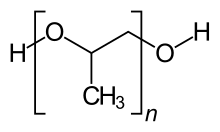
 | |
| Names | |
|---|---|
| IUPAC name | |
| Identifiers | |
| |
| ECHA InfoCard | 100.105.547 |
| UNII | |
CompTox Dashboard (EPA) |
|
| Properties | |
| C3nH6n+2On+1 | |
| Molar mass | 58.08n + 18.08 (repeat unit), mass of polymer variable |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Polypropylene glycol or polypropylene oxide is the polymer (or macromolecule) of propylene glycol.[1] Chemically it is a polyether, and, more generally speaking, it's a polyalkylene glycol (PAG) H S Code 3907.2000. The term polypropylene glycol or PPG is reserved for polymer of low- to medium-range molar mass when the nature of the end-group, which is usually a hydroxyl group, still matters. The term "oxide" is used for high-molar-mass polymer when end-groups no longer affect polymer properties. Between 60 and 70% of propylene oxide is converted to polyether polyols by the process called alkoxylation.[2]
Polymerization
Polypropylene glycol is produced by ring-opening polymerization of propylene oxide. The initiator is an alcohol and the catalyst a base, usually potassium hydroxide. When the initiator is ethylene glycol or water the polymer is linear. With a multifunctional initiator like glycerine, pentaerythritol or sorbitol the polymer branches out.
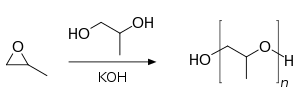
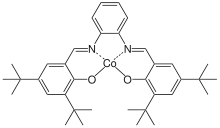
Conventional polymerization of propylene oxide results in an atactic polymer. The isotactic polymer can be produced from optically active propylene oxide, but at a high cost. A salen cobalt catalyst was reported in 2005 to provide isotactic polymerization of the prochiral propylene oxide[3]
Properties
PPG has many properties in common with polyethylene glycol. The polymer is a liquid at room temperature. Solubility in water decreases rapidly with increasing molar mass. Secondary hydroxyl groups in PPG are less reactive than primary hydroxyl groups in polyethylene glycol. PPG is less toxic than PEG, so biotechnologicals are now mainly produced with PPG.[4][5][6]

Uses
PPG is used in many polyurethane formulations.[7][8] Synthesis of waterborne polymers has been a feature with this substance.[9] As the basic building block is propylene oxide, there are 3 carbons per oxygen on the backbone. This confers some degree of water miscibility though not as good as ethylene oxide based molecules.[10] It is used to synthesize the epoxy reactive diluent and flexibilizer, Poly(propylene glycol) diglycidyl ether.[11][12] Another use of PPG is as a surfactant, wetting agent and dispersant in leather finishing.[13] PPG is also employed as a reference and calibrant in mass spectrometry and HPLC.[14][15] PPG and derivatives maybe used as defoamers in drilling and other applications.[16][17] It is also used as a primary ingredient in the making of paintballs.[18] It has been evaluated as a corrosion inhibitor.[19]
References
- ↑ "2-(2-Hydroxypropoxy)propan-1-ol". PubChem. U.S. National Library of Medicine. Retrieved 2022-11-14.
- ↑ Adam N, Avar G, Blankenheim H, Friederichs W, Giersig M, Weigand E, et al. "Polyurethanes". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a21_665.pub2. ISBN 978-3527306732.
- ↑ Peretti KL, Ajiro H, Cohen CT, Lobkovsky EB, Coates GW (August 2005). "A highly active, isospecific cobalt catalyst for propylene oxide polymerization". Journal of the American Chemical Society. 127 (33): 11566–7. doi:10.1021/ja053451y. PMID 16104709.
- ↑ Thurman EM, Ferrer I, Rosenblum J, Linden K, Ryan JN (February 2017). "Identification of polypropylene glycols and polyethylene glycol carboxylates in flowback and produced water from hydraulic fracturing". Journal of Hazardous Materials. Special Issue on Emerging Contaminants in engineered and natural environment. 323 (Pt A): 11–17. doi:10.1016/j.jhazmat.2016.02.041. PMID 26947804.
- ↑ Wang R, Hughes T, Beck S, Vakil S, Li S, Pantano P, Draper RK (November 2013). "Generation of toxic degradation products by sonication of Pluronic® dispersants: implications for nanotoxicity testing". Nanotoxicology. 7 (7): 1272–1281. doi:10.3109/17435390.2012.736547. PMC 3657567. PMID 23030523.
- ↑ Thangavelu SA, Mukherjee M, Layana K, Kumar CD, Sulthana YR, Kumar RR, et al. (2020-06-15). "Biodegradable polyurethanes foam and foam fullerenes nanocomposite strips by one-shot moulding: Physicochemical and mechanical properties". Materials Science in Semiconductor Processing. 112: 105018. doi:10.1016/j.mssp.2020.105018. ISSN 1369-8001. S2CID 213206957.
- ↑ Chattopadhyay DK, Raju NP, Vairamani M, Raju KV (2008-04-01). "Structural investigations of polypropylene glycol (PPG) and isophorone diisocyanate (IPDI) based polyurethane prepolymer by matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF)-mass spectrometry". Progress in Organic Coatings. 62 (2): 117–122. doi:10.1016/j.porgcoat.2007.09.021. ISSN 0300-9440.
- ↑ Xu Z, Cui Y, Li T, Dang H, Li J, Cheng F (July 2020). "Enhanced Mechanical and Shape Memory Properties of Poly(propylene glycol)‐Based Star‐Shaped Polyurethane". Macromolecular Chemistry and Physics. 221 (13): 2000082. doi:10.1002/macp.202000082. ISSN 1022-1352. S2CID 225563251.
- ↑ US 10920011, Kim KJ, Hu R, Grove JL, "Water soluble polymers and polymer adducts along with aqueous solutions thereof Patent", issued 16 February 2021, assigned to Arkema Inc
- ↑ Howarth GA (April 1997). Synthesis of a legislation compliant corrosion protection coating system based on urethane, oxazolidine and waterborne epoxy technology (Master of Science thesis). London: Imperial College. pp. 36–28, 52–60.
- ↑ Crivello JV (2006). "Design and synthesis of multifunctional glycidyl ethers that undergo frontal polymerization". Journal of Polymer Science Part A: Polymer Chemistry. 44 (21): 6435–6448. Bibcode:2006JPoSA..44.6435C. doi:10.1002/pola.21761. ISSN 0887-624X.
- ↑ US 5162547, Roth M, Wolleb H, Truffer MA, "Process for the preparation of glycidyl ethers", published 1992-11-10, assigned to Ciba-Geigy Corp.
- ↑ Zgoła-Grześkowiak A, Grześkowiak T, Zembrzuska J, Łukaszewski Z (July 2006). "Comparison of biodegradation of poly(ethylene glycol)s and poly(propylene glycol)s". Chemosphere. 64 (5): 803–809. Bibcode:2006Chmsp..64..803Z. doi:10.1016/j.chemosphere.2005.10.056. PMID 16343594.
- ↑ Rychłowska J, Zgoła A, Grześkowiak T, Lukaszewski Z (December 2003). "Isolation of poly(propylene glycol)s from water for quantitative analysis by reversed-phase liquid chromatography". Journal of Chromatography A. 1021 (1–2): 11–17. doi:10.1016/j.chroma.2003.09.003. PMID 14735971.
- ↑ Castillo M, Ventura F, Barceló D (1999-04-01). "Sequential solid phase extraction protocol followed by liquid chromatography–atmospheric pressure chemical ionization–mass spectrometry for the trace determination of non ionic polyethoxylated surfactants in tannery wastewaters". Waste Management. 19 (2): 101–110. doi:10.1016/S0956-053X(99)00004-5. ISSN 0956-053X.
- ↑ Tan SN, Pugh RJ, Fornasiero D, Sedev R, Ralston J (February 2005). "Foaming of polypropylene glycols and glycol/MIBC mixtures ]". Minerals Engineering. Reagents '04. 18 (2): 179–188. doi:10.1016/j.mineng.2004.08.017. ISSN 0892-6875.
- ↑ Bava L, Mahmoudkhani A, Wilson R, Levy L (2013-03-23). "New Generation of "Green" Defoamers for Challenging Drilling and Cementing Applications". New Generation of Green Defoamers for Challenging Drilling and Cementing Applications. OnePetro. doi:10.2118/164504-MS.
- ↑ Dhiman M, Singh K, Gupta DP, Singh DP, Upmanyu A (2020-05-04). "Study of excess acoustical and thermo-dynamical parameters of binary solutions of polypropylene glycol-400 and n-alkanols at 303 K". AIP Conference Proceedings. 2220 (1): 080001. doi:10.1063/5.0001107. ISSN 0094-243X. S2CID 218923617.
- ↑ Solomon MM, Umoren SA, Israel AU, Etim IG (January 2016). "Synergistic inhibition of aluminium corrosion in H2SO4 solution by polypropylene glycol in the presence of iodide ions". Pigment & Resin Technology. 45 (4): 280–293. doi:10.1108/PRT-01-2015-0010. ISSN 0369-9420.