 | |
| Names | |
|---|---|
| Preferred IUPAC name
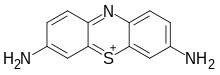
3,7-Diamino-5λ4-phenothiazin-5-ylium | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.008.611 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C12H10N3S+ | |
| Molar mass | 228.29 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Thionine, also known as Lauth's violet, is the salt of a heterocyclic compound. It was firstly synthesised by Charles Lauth. A variety of salts are known including the chloride and acetate, called respectively thionine chloride and thionine acetate. The dye is structurally related to methylene blue, which also features a phenothiazine core.[1] The dye's name is frequently misspelled with omission of the e, and is not to be confused with the plant protein thionin. The -ine ending indicates that the compound is an amine.[2][3]
Dye properties and use
Thionine is a strongly staining metachromatic dye, which is widely used for biological staining.[4] Thionine can also be used in place of Schiff reagent in quantitative Feulgen staining of DNA. It can also be used to mediate electron transfer in microbial fuel cells.[5] Thionine is a pH-dependent redox indicator with E0 = 0.06 at pH 7.0. Its reduced form, leuco-thionine, is colorless.
When both the amines are dimethylated, the product tetramethyl thionine is the famous methylene blue, and the intermediates are Azure C (Monomethyl thionine), Azure A (when one of the amines is dimethylated and the other remains a primary amine), and Azure B (Trimethyl thionine). When methylene blue is "polychromed" by ripening (oxidized in solution or metabolized by fungal contamination,[6] as originally noted in the thesis of Dr D L Romanowski in 1890s), it forms thionine and all the Azure intermediates.[7][8]
Notes and references
- ↑ Gérard Taurand, "Phenothiazine and Derivatives" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005.doi:10.1002/14356007.a19_387
- ↑ Kiernan JA (2001). "Classification and naming of dyes, stains and fluorochromes". Biotech Histochem. 76 (5–6): 261–78. doi:10.1080/bih.76.5-6.261.278. PMID 11871748. S2CID 32479873.
- ↑ Webster's Third New International Dictionary. G & C Merriam Co. 1976, p.2377.
- ↑ "Stainsfile — Thionin".
- ↑ Eugenii Katz; Andrew N. Shipway; Itamar Willner (2003). "21". In Wolf Vielstich (ed.). Handbook of Fuel Cells: Fundamentals, Technology, Applications, 4-Volume Set (PDF). Wiley. p. 5. ISBN 978-0-471-49926-8.
- ↑ Dako Education Guide - Special Stains and H & E ” second edition Chapter 19: On Chemical Reactions and Staining Mechanisms by John A. Kiernan, Subsection What is Giemsa’s stain and how does it color blood cells, bacteria and chromosomes? p172 Archived 2012-05-13 at the Wayback Machine
- ↑ Wilson TM (1907). "On the Chemistry and Staining Properties of Certain Derivatives of the Methylene Blue Group when Combined with Eosin". J Exp Med. 9 (6): 645–70. doi:10.1084/jem.9.6.645. PMC 2124692. PMID 19867116.
- ↑ Marshall, PN (1978) Romanowsky-type stains in haematology. Histochemical Journal 10: 1-29.