 | |
| Names | |
|---|---|
| IUPAC name
(2S,4aR,6aR,7S,7aS,8aS,9S,9aS,9bS)-2-(3-Furanyl)dodecahydro-7-hydroxy-6a,9b-dimethyl-9,7-(epoxymethano)-4H-oxireno[6,7]naphtho[2,1-c]pyran-4,11-dione | |
| Other names
2,3-Epoxycolumbin; Jateorin; 5-(furan-3-yl)-12-hydroxy-3,11-dimethyl-6,14,16-trioxapentacyclo[10.3.2.02,11.03,8.013,15]heptadecane-7,17-dione | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C20H22O7 | |
| Molar mass | 374.389 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Tinosporide is a chemical compound classified as a diterpenoid and a furanolactone. It was first isolated from the plant Tinospora cordifolia, from which it derives its name.[1][2] It has since been found in other plants of the genus Tinospora, such as Tinospora glabra.
Because Tinospora cordifolia has been used in traditional herbal medicine, there has been research directed at exploring the potential pharmacology of tinosporide and related compounds.[3]
Related compounds
Other diterpenoid furanolactones with a similar structure include columbin, palmarin, and chasmanthin.
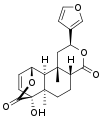 Columbin
Columbin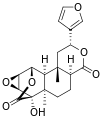 Palmarin
Palmarin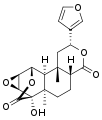 Chasmanthin
Chasmanthin
External links
- ↑ Swaminathan, K.; Sinha, U. C.; Bhatt, R. K.; Sabata, B. K.; Tavale, S. S. (1989). "Structure of tinosporide, a diterpenoid furanolactone from Tinospora cordifolia Miers". Acta Crystallographica Section C. 45 ( Pt 1): 134–136. doi:10.1107/s0108270188009953. PMID 2610955.
- ↑ Sharma, Priyanka; Dwivedee, Bharat P.; Bisht, Dheeraj; Dash, Ashutosh K.; Kumar, Deepak (2019). "The chemical constituents and diverse pharmacological importance of Tinospora cordifolia". Heliyon. 5 (9): e02437. Bibcode:2019Heliy...502437S. doi:10.1016/j.heliyon.2019.e02437. PMC 6827274. PMID 31701036.
- ↑ Pathak, Ashish K.; Jain, Dharam C.; Sharma, Ram P. (1995). "Chemistry and Biological Activities of the Genera Tinospora". International Journal of Pharmacognosy. 33 (4): 277–287. doi:10.3109/13880209509065379.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.