| |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /taɪˈzænɪdiːn/ tye-ZAN-i-deen |
| Trade names | Zanaflex, Sirdalud, and others |
| Other names | 4-Chloro-N-(4,5-dihydro-1H-imidazol-2-yl)-8-thia-7,9-diazabicyclo[4.3.0]nona-2,4,6,9-tetraen-5-amine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601121 |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | ~40%[1] |
| Protein binding | ~30% |
| Metabolism | Liver (CYP1A2, 95%) |
| Elimination half-life | 2.54 hours (tizanidine), 20–40 hours (inactive metabolites)[1] |
| Excretion | Urine (60%), feces (20%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.125.400 |
| Chemical and physical data | |
| Formula | C9H8ClN5S |
| Molar mass | 253.71 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Tizanidine, sold under the brand name Zanaflex among others, is an alpha-2 (α2) adrenergic receptor agonist,[2] similar to clonidine, that is used to treat muscle spasticity due to spinal cord injury, multiple sclerosis, and spastic cerebral palsy.[3] Effectiveness appears similar to baclofen or diazepam.[4] It is taken by mouth.[5]
Common side effects of tizanidine include dry mouth, sleepiness, weakness, and dizziness.[5] Serious side effects may include low blood pressure, liver problems, psychosis, and QT prolongation.[5] It is unclear if use in pregnancy and breastfeeding is safe.[6] It is an α2-adrenergic agonist, but how it works is not entirely clear.[5]
Tizanidine was approved for medical use in the United States in 1996.[5] It is available as a generic medication.[3] In 2020, it was the 84th most commonly prescribed medication in the United States, with more than 8 million prescriptions.[7][8]
Medical uses
Tizanidine has been found to be as effective as other antispasmodic drugs and is more tolerable than baclofen and diazepam.[4]
Side effects
Side effects include dizziness, drowsiness, weakness, nervousness, confusion, hallucinations, strange dreams, depression, vomiting, dry mouth, constipation, diarrhea, stomach pain, heartburn, increased muscle spasms, back pain, rash, sweating, and a tingling sensation in the arms, legs, hands, and feet.[9]
Symptoms of overdose in 45 cases reported to a poison control center included: lethargy, bradycardia, hypotension, agitation, confusion, vomiting and coma.[10]
Tizanidine has drug abuse potential. It has been recognized to cause dependence and addiction.[11] In the US it is not regulated under the Controlled Substances Act, however.
Interactions
Concomitant use of tizanidine and moderate or potent CYP1A2 inhibitors (such as zileuton, certain antiarrhythmics (amiodarone, mexiletine, propafenone, verapamil), cimetidine, famotidine, aciclovir, ticlopidine and oral contraceptives) is contraindicated. Concomitant use of tizanidine with fluvoxamine, a potent CYP1A2 inhibitor in humans, resulted in a 33-fold increase in the tizanidine AUC (plasma drug concentration-time curve).[1] For this reason both fluvoxamine and tizanidine should not be taken at the same time. Fluoroquinolone antibiotics such as moxifloxacin, levofloxacin, and ciprofloxacin should also be avoided due to an increased serum concentration of tizanidine when administered concomitantly.[12] Tizanidine has the potential to interact with other central nervous system depressants. Alcohol should be avoided, particularly as it can upset the stomach. The CNS-depressant effects of tizanidine and alcohol are additive.[1] Caution with the following interactions:[13][14][15]
- antibiotics such as enoxacin, gatifloxacin, levofloxacin, lomefloxacin, moxifloxacin, ofloxacin, sparfloxacin, trovafloxacin, or norfloxacin;
- blood pressure medications such as clonidine, guanabenz, guanfacine (Tenex), or methyldopa;
- heart rhythm medications such as amiodarone (Cordarone, Pacerone), mexiletine (Mexitil), propafenone (Rhythmol), and verapamil (Calan, Covera, Isoptin).
Pharmacology
Tizanidine is an α2 receptor agonist closely related to clonidine. It has approximately one tenth to one fifteenth of the blood pressure lowering effect of clonidine. The relation between the α2 receptor agonism and the spasmolytic action is still not fully understood.[2]
| Site | Ki (nM) | Species | Ref |
|---|---|---|---|
| α2A | 62 | Human | [16] |
| α2B | 75 | OK | [16] |
| α2C | 76 | Rat | [16] |
Route of administration
Tizanidine is available as a tablet or capsule. Capsules may be opened and sprinkled on food. However, this may change the absorption of the medication compared to taking the capsule whole.[17] It has a volume of distribution of 2.4 L/kg following intravenous administration.[1]
Chemistry
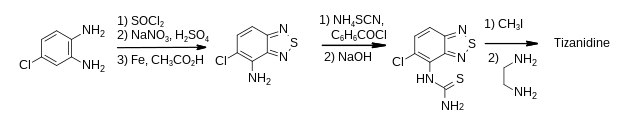
Tizanidine is a derivative of 2,1,3-benzothiadiazole and its first published synthesis was reported in a patent.[18] The 5-chloro-2,1,3-benzothiadiazol-4-amine intermediate was a known compound, produced in three steps from 4-chlorophenylenediamine as shown.[19] Treatment with two equivalents of thionyl chloride in pyridine formed the heterocycle, which was nitrated with sodium nitrate in sulfuric acid and reduced using iron and acetic acid.
The amine intermediate was treated with benzoyl chloride and ammonium thiocyanate followed by alkaline hydrolysis to form a thiourea. This was activated as its isothiuronium salt before being treated with ethylene diamine to give tizanidine.[18][20]
See also
- Xylazine, an alpha-2 (α2) adrenergic receptor agonist and clonidine analogue used in veterinary medicine and as a street drug
References
- 1 2 3 4 5 "Zanaflex (tizanidine hydrochloride) Capsules and Tablets for Oral Use. Full Prescribing Information" (PDF). Acorda Therapeutics Inc. Ardsley, NY 10502. Retrieved 1 September 2016.
- 1 2 Katzung BG (30 November 2017). Basic & clinical pharmacology (14th ed.). New York: McGraw Hill Education. p. 487. ISBN 9781259641152. OCLC 1015240036.
- 1 2 British national formulary: BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 1094. ISBN 9780857113382.
- 1 2 Kamen L, Henney HR, Runyan JD (February 2008). "A practical overview of tizanidine use for spasticity secondary to multiple sclerosis, stroke, and spinal cord injury". Current Medical Research and Opinion. 24 (2): 425–439. doi:10.1185/030079908X261113. PMID 18167175. S2CID 73086671.
- 1 2 3 4 5 "Tizanidine Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 3 March 2019.
- ↑ "Tizanidine Pregnancy and Breastfeeding Warnings". Drugs.com. Retrieved 3 March 2019.
- ↑ "The Top 300 of 2020". ClinCalc. Retrieved 7 October 2022.
- ↑ "Tizanidine - Drug Usage Statistics". ClinCalc. Retrieved 7 October 2022.
- ↑ "Page not available". PubMed Health. Archived from the original on 11 November 2012. Retrieved 4 September 2017.
- ↑ Spiller HA, Bosse GM, Adamson LA (January 2004). "Retrospective review of Tizanidine (Zanaflex) overdose". Journal of Toxicology. Clinical Toxicology. 42 (5): 593–596. doi:10.1081/CLT-200026978. PMID 15462150.
- ↑ Haukka J, Kriikku P, Mariottini C, Partonen T, Ojanperä I (March 2018). "Non-medical use of psychoactive prescription drugs is associated with fatal poisoning". Addiction. 113 (3): 464–472. doi:10.1111/add.14014. PMID 28841781.
- ↑ "Tizanidine Uses, Dosage, Side Effects – Drugs.com". drugs.com. Retrieved 4 September 2017.
- ↑ NHS Wales (2011). "Tizanidine (Zanaflex) Gwent Primary Care Prescribing Guidance" (PDF). Aneurin Bevan University Health Board.
- ↑ "Tizanidine package leaflet: Information for the user" (PDF). Medicines and Healthcare products Regulatory Agency (UK). 2016. Archived from the original (PDF) on 16 September 2018. Retrieved 16 September 2018.
- ↑ "Zanaflex (tizanidine hydrochloride) dose, indications, adverse effects, interactions... from PDR.net". www.pdr.net. Retrieved 16 September 2018.
- 1 2 3 Blaxall HS, Murphy TJ, Baker JC, Ray C, Bylund DB (October 1991). "Characterization of the alpha-2C adrenergic receptor subtype in the opossum kidney and in the OK cell line". The Journal of Pharmacology and Experimental Therapeutics. 259 (1): 323–329. PMID 1656026.
- ↑ "Tizanidine: MedlinePlus Drug Information". medlineplus.gov. U.S. National Library of Medicine. Retrieved 29 October 2019.
- 1 2 US patent 3843668, Neumann P, "Certain 4-substituted amino-2,1,3-benzothiadiozoles", published 1974-10-22, assigned to Sandoz AG
- ↑ Pesin VG, Sergeev VA (1969). "Research on 2,1,3-thia- and selenadiazole". Chemistry of Heterocyclic Compounds. 3 (5): 662–666. doi:10.1007/BF00468340. S2CID 98830770.
- ↑ "Pharmaceutical Substances: Tizanidine". Thieme. Retrieved 16 February 2023.
External links
- "Tizanidine". Drug Information Portal. U.S. National Library of Medicine.
- "Tizanidine hydrochloride". Drug Information Portal. U.S. National Library of Medicine.
- "Tizanidine side effects". Drug Information Portal. tizanidine-side-effects.us. Archived from the original on 28 December 2021. Retrieved 17 December 2020.