 | |
| Names | |
|---|---|
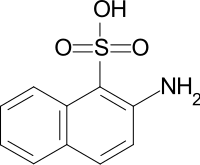
| Preferred IUPAC name
2-Aminonaphthalene-1-sulfonic acid | |
| Other names
2-Naphthylamine-1-sulfonic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.211 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H9NO3S | |
| Molar mass | 223.25 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Tobias acid (2-amino-1-naphthalenesulfonic acid) is an organic compound with the formula C10H6(SO3H)(NH2). It is named after the German chemist Georg Tobias.[1] It is one of several aminonaphthalenesulfonic acids, which are derivatives of naphthalene containing both amine and sulfonic acid functional groups. It is a white solid, although commercial samples can appear otherwise.[2] It is used in the synthesis of azo dyes such as C.I. Acid Yellow 19 and C.I. Pigment Red 49. It is prepared via the Bucherer reaction of 2-hydroxynaphthalene-1-sulfonic acid with ammonia and ammonium sulfite.[3]
References
- ↑ Tobias, Georg (January 1890). "Zur Anwendung der Sandmeyer' schen Reaction auf Diazosulfosäuren und über die Zersetzung dieser Verbindungen bei Gegenwart von Kupfer oder Kupferoxydul" [On the application of Sandmeyer's reaction to diazosulfonic acids and the decomposition of these compounds in the presence of copper or copper oxide]. Berichte der Deutschen Chemischen Gesellschaft (in German). 23 (1): 1628–1634. doi:10.1002/cber.189002301270.
- ↑ CID 6670 from PubChem
- ↑ Gerald Booth "Naphthalene Derivatives" in Ullmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a17_009.
External links
- 2-Amino-1-naphthalenesulfonic acid, NIST Standard Reference Data Program
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.