 | |
| Names | |
|---|---|
| Preferred IUPAC name
Hexamethyl-1,3,5-trithiane | |
| Other names
Trithioacetone[1] | |
| Identifiers | |
3D model (JSmol) |
|
| 5-19-09-00119 | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.011.438 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C9H18S3 | |
| Molar mass | 222.42 g·mol−1 |
| Density | 1.0660 to 1.0700 g/mL[2] |
| Melting point | 21.8°C[3] |
| Boiling point | 107°C/10mmHg[2] |
Refractive index (nD) |
1.5390 to 1.5430[2] |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
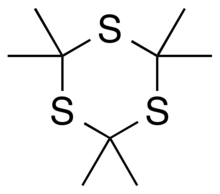
Trithioacetone (2,2,4,4,6,6-hexamethyl-1,3,5-trithiane) is an organic chemical with formula C
9H
18S
3. Its covalent structure is [–C(CH
3)
2–S–]
3, that is, a six-membered ring of alternating carbon and sulfur atoms, with two methyl groups attached to each carbon.[4][2] It can be viewed as a derivative of 1,3,5-trithiane, with methyl-group substituents for all of the hydrogen atoms in that parent structure.
The compound Trithioacetone is a stable cyclic trimer of thioacetone (propane-2-thione), which by itself is an unstable compound.[5][6] In contrast, the analogous trioxane compound, 2,2,4,4,6,6-hexamethyl-1,3,5-trioxane (Triacetone), with oxygen atoms in place of the sulfur atoms, seems to be unstable, while its corresponding monomer acetone (2-propanone) is stable.
Synthesis
Trithioacetone was first made in 1889 by Baumann and Fromm, by reaction of hydrogen sulfide with acetone.[6] In the presence of an acidified ZnCl
2 catalyst at 25 °C, one obtains a product that is 60–70% trithioacetone, 30–40% of 2,2-propanedithiol, and small amounts of two isomeric impurities, 3,3,5,5,6,6-hexamethyl 1,2,4-trithiane and 4-mercapto-2,2,4,6,6-pentamethyl-1,3-dithiane.[6] The product can also be obtained by pyrolysis of allyl isopropyl sulfide.[7][8]
Reactions
Pyrolysis of trithioacetone at 500–650 °C and 5–20 mm of Hg gives thioacetone, that can be collected by a cold trap at −78 °C.

Uses
Trithioacetone is found in some flavoring agents. Its FEMA number is 3475.[9][10][11][12]
Toxicity
See also
- 2,4,6-trimethyl-1,3,5-trithiane
- Hexamethylcyclotrisiloxane, an analog with a silicon-oxygen ring instead of a carbon-sulfur one.
- Hexamethylcyclotrisilazane, with a silicon-nitrogen ring.[13]
- 2,2,4,4,6,6-hexamethyl-1,3,5-triselena-2,4,6-tristannacyclohexane, with a tin-selenium ring.[14][15]
References
- ↑ "Trithioacetone". Online chemical data sheet, accessed on 2020-01-01.
- 1 2 3 4 TCI America (2020): "Product H1278: 2,2,4,4,6,6-Hexamethyl-1,3,5-trithiane". Online catalog page, accessed on 2020-01-01.
- 1 2 NCBI PubChem (2010): "2,2,4,4,6,6-Hexamethyl-1,3,5-trithiane". Online chemical data sheet, accessed on 2020-01-01.
- ↑ David S. Breslow, Herman Skolnik (2009): Multi-Sulfur and Sulfur and Oxygen Five- and Six-Membered Heterocycles, Part 2; page 712. Volume 68 of Chemistry of Heterocyclic Compounds. ISBN 9780470188330
- ↑ R. D. Lipscomb and W. H. Sharkey (1970): "Characterization and polymerization of thioacetone". Journal of Polymer Science – Part A: Polymer Chemistry, volume 8, issue 8, pages 2187–2196. doi:10.1002/pol.1970.150080826
- 1 2 3 William H. Sharkey (1979): "Polymerization through the carbon-sulfur double bond". Polymerization, series Advances in Polymer Science, volume 17, pages 73–103. doi:10.1007/3-540-07111-3_2
- ↑ William J. Bailey and Hilda Chu (1965): "Synthesis of polythioacetone". ACS Polymer Preprints, volume 6, pages=145–155
- ↑ Horst Bohme, Hans Pfeifer, and Erich Schneider (1942): "Dimeric thioketones". Berichte der Deutschen Chemischen Gesellschaft, volume 75B, issue 7, pages 900–909. doi:10.1002/cber.19420750722 Note: This early report mistakes the trimer for the monomer.
- 1 2 E. J. Moran, O. D. Easterday, and B. L. Oser (1980): "Acute oral toxicity of selected flavor chemicals". Drug and Chemical Toxicology, volume 3, issue 3, pages 249–258.PMID 7449655 doi:10.3109/01480548009002221
- ↑ World Health Organization (1999): "Trithioacetone". Online data sheet in the Evaluation of the Joint FAO/WHO Expert Committee on Food Additives (JECFA). Accessedd on 2020-01-02.
- ↑ G. Ohloff and I. Flament (1979): "The Role of Heteroatomic Substances in the Aroma Compounds of Foodstuffs". In Fortschritte der Chemie Organischer Naturstoffe (Progress in the Chemistry of Organic Natural Products), volume 36, pages 231–283. doi:10.1007/978-3-7091-3265-4_2
- ↑ EUR-Lex (2012): "Table entry 15.009: Trithioacetone". In EU Regulation No. 872/2012, Document 32012R0872, Official Journal of the EU – Series L, volume 267, pages 1–161.
- ↑ Stuart D. Brewer and Charles P. Haber (1948): "Alkylsilazanes and Some Related Compounds". Journal of the American Chemical Society, volume 70, issue 11, pages 3888–3891. doi:10.1021/ja01191a106
- ↑ B. M. Mikhova (2008), "NMR Data for Carbon-13 – C6H18Se3Sn3" in Landolt-Börnstein – Group III Condensed Matter, volume 35 Nuclear Magnetic Resonance Data, subvolume D5, Organometallic Compounds. doi:10.1007/978-3-540-74189-3_1362
- ↑ Martin Dräger, Axel Blecher, Hans-Jürgen Jacobsen, Bernt Krebs (1978): "Molekül- und kristallstruktur von hexamethylcyclo-tristannaselenan [(CH
3)
2SnSe]
3". Journal of Organometallic Chemistry, volume 161, issue 3, pages 319–325. doi:10.1016/S0022-328X(00)92243-5