 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
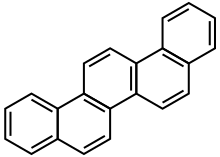
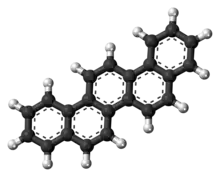
Picene[2] | |
| Other names
Dibenzo[a,i]phenanthrene 3,4-Benzchrysene β,β-Binaphthylene ethene | |
| Identifiers | |
3D model (JSmol) |
|
| 1912414 | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.005.381 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C22H14 | |
| Molar mass | 278.33 g/mol |
| Density | ? g/cm3 |
| Melting point | 366 to 367 °C (691 to 693 °F; 639 to 640 K) |
| Boiling point | 518 to 520 °C (964 to 968 °F; 791 to 793 K) |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H371 | |
| P260, P264, P270, P309+P311, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Picene is a hydrocarbon found in the pitchy residue obtained in the distillation of peat tar and of petroleum. This is distilled to dryness and the distillate repeatedly recrystallized from cymene. It may be synthetically prepared by the action of anhydrous aluminium chloride on a mixture of naphthalene and 1,2-dibromoethane, or by distilling a-dinaphthostilbene. It crystallizes in large colorless plates which possess a blue fluorescence. It is soluble in concentrated sulfuric acid with a green color. Chromic acid in glacial acetic acid solution oxidizes it to picene-quinone, picene-quinone carboxylic acid, and finally to phthalic acid.
When intercalated with potassium or rubidium and cooled to below 18 K, picene has been reported to exhibit superconductive properties.[3] However, due to the apparent inability to reproduce this work,[4] the superconducting nature of doped picene has been met with heavy scepticism.[5]
Picene is also a major constituent of the hydrocarbon mineral idrialite.
See also
- Olympicene, which has the same number of rings linked in a different way
References
- ↑ Merck Index, 11th Edition, 7368.
- ↑ Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 206. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ↑ Das, Saswato (March 2010), "Hydrocarbon Superconductor Discovered", IEEE Spectrum
- ↑ Artioli, Gianluca A.; Malavasi, Lorenzo (Dec 2013), "Superconductivity in metal-intercalated aromatic hydrocarbons", J. Mater. Chem. C, 2 (9): 1577, doi:10.1039/C3TC32326A
- ↑ Heguri, Satoshi; Kobayashi, Mototada; Tanigaki, Katsumi (May 2015), "Questioning the existence of superconducting potassium doped phases for aromatic hydrocarbons", Phys. Rev. B, 92 (1): 014502, Bibcode:2015PhRvB..92a4502H, doi:10.1103/PhysRevB.92.014502
This article incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). "Picene". Encyclopædia Britannica. Vol. 21 (11th ed.). Cambridge University Press. p. 581.