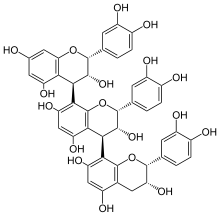
 | |
| Names | |
|---|---|
| IUPAC name
[(2R,3R,4R)-Flavan-3,3′,4′,5,7-pentol]-(4→8)-[(2R,3R,4S)-flavan-3,3′,4′,5,7-pentol]-(4→8)-[(2R,3R)-flavan-3,3′,4′,5,7-pentol] | |
| Systematic IUPAC name
(12R,13R,14R,22R,23R,24S,32R,33R)-12,22,32-Tris(3,4-dihydroxyphenyl)-12H,22H,32H-[14,28:24,38-ter-1-benzopyran]-13,15,17,23,25,27,33,35,37-nonol | |
| Other names
Procyanidin C1 Procyanidol C1 Epicatechin-(4.beta.-->8)epicatechin-(4.beta.-->8)epicatechin Epicatechin-(4β→8)-epicatechin--(4β→8)-epicatechin | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C45H38O18 | |
| Molar mass | 866.77 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Procyanidin C1 (PCC1) is a B type proanthocyanidin. It is an epicatechin trimer found in grape (Vitis vinifera),[1] unripe apples,[2] and cinnamon.[3]
Natural occurrence and function
Procyanidin C1 can be isolated from grape seed extract.[4]
Chemical synthesis
The stereoselective synthesis of seven benzylated proanthocyanidin trimers (epicatechin-(4β-8)-epicatechin-(4β-8)-epicatechin trimer (procyanidin C1), catechin-(4α-8)-catechin-(4α-8)-catechin trimer (procyanidin C2), epicatechin-(4β-8)-epicatechin-(4β-8)-catechin trimer and epicatechin-(4β-8)-catechin-(4α-8)-epicatechin trimer derivatives) can be achieved with TMSOTf-catalyzed condensation reaction, in excellent yields. The structure of benzylated procyanidin C2 was confirmed by comparing the 1H NMR spectra of protected procyanidin C2 that was synthesized by two different condensation approaches. Finally, deprotection of (+)-catechin and (-)-epicatechin trimers derivatives gives four natural procyanidin trimers in good yields.[5]
Research
Procyanidin C1 has been shown to be an effective senolytic agent in wild-type mice, with effects of increased lifespan, reduced senescence markers and making them fitter. It was also found to greatly increase the effectiveness of chemotherapy in mice in which human prostate tumor cells were implanted.[4][6]
See also
External links
- Carissa Wong: Grape seed chemical allows mice to live longer by killing aged cells. NewScientist, 6 December 2021
References
- ↑ Proanthocyanidin composition of red Vitis vinifera varieties from the Douro valley during ripening : Influence of cultivation altitude. Mateus Nuno, Marques Sara, Goncalves Ana C., Machado José M. and De Freitas Victor, American journal of enology and viticulture, 2001, vol. 52, no2, pp. 115-121, INIST 1129642
- ↑ Nakano, Nobuhiro; Nishiyama, Chiharu; Tokura, Tomoko; Nagasako-Akazome, Yoko; Ohtake, Yasuyuki; Okumura, Ko; Ogawa, Hideoki (2008-07-02). "Procyanidin C1 from Apple Extracts Inhibits FcεRI-Mediated Mast Cell Activation". International Archives of Allergy and Immunology. 147 (3): 213–221. doi:10.1159/000142044. ISSN 1018-2438. PMID 18594151. S2CID 43866026. Retrieved 2022-02-20.
- ↑ Sun, Peng; Li, Kai; Wang, Ting; Ji, Jun; Wang, Yan; Chen, Kai-Xian; Jia, Qi; Li, Yi-Ming; Wang, He-Yao (2019-07-23). "Procyanidin C1, a Component of Cinnamon Extracts, Is a Potential Insulin Sensitizer That Targets Adipocytes". Journal of Agricultural and Food Chemistry. American Chemical Society (ACS). 67 (32): 8839–8846. doi:10.1021/acs.jafc.9b02932. ISSN 0021-8561. PMID 31334651. S2CID 198169719.
- 1 2 Carissa Wong (Dec 6, 2021). "Grape seed chemical allows mice to live longer by killing aged cells". New Scientist.
- ↑ Efficient Stereoselective Synthesis of Proanthocyanidin Trimers with TMSOTf-Catalyzed Intermolecular Condensation. Akiko Saito, Akira Tanaka, Makoto Ubukata and Noriyuki Nakajima, Synlett, 2004, volume 6, pages 1069-1073, doi:10.1055/s-2004-822905
- ↑ Xu, Qixia; et al. (Dec 6, 2021). "The flavonoid procyanidin C1 has senotherapeutic activity and increases lifespan in mice". Nature Metabolism. 3 (12): 1706–1726. doi:10.1038/s42255-021-00491-8. PMC 8688144. PMID 34873338.