 | |
 | |
| Legal status | |
|---|---|
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
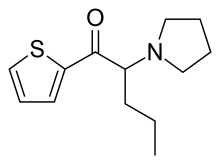
| Formula | C13H19NOS |
| Molar mass | 237.36 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
α-Pyrro
α-PVT was first identified in Japan in 2013.[4] Its metabolism has been described in literature.[5][6]
Side effects
α-PVT has been shown to possess high cytotoxicity against human cell lines.[7]
Legality
Sweden's public health agency suggested classifying α-PVT as hazardous substance on November 10, 2014.[8]
As of October 2015 α-PVT is a controlled substance in China.[9]
α-PVT is illegal in Switzerland as of December 2015.[10]
See also
- α-Pyrrolidinopropiophenone (α-PPP)
- α-Pyrrolidinobutiophenone (α-PBP)
- α-Pyrrolidinohexiophenone (α-PHP)
- Methiopropamine
- Naphyrone (O-2482)
- Pyrovalerone (O-2371)
- Thiopropamine
- Thiothinone
References
- ↑ "α-Pyrrolidinopentiothiophenone". Cayman Chemical. Retrieved 29 June 2015.
- ↑ Concheiro M, Castaneto M, Kronstrand R, Huestis MA (June 2015). "Simultaneous determination of 40 novel psychoactive stimulants in urine by liquid chromatography-high resolution mass spectrometry and library matching". Journal of Chromatography. A. 1397 (5): 32–42. doi:10.1016/j.chroma.2015.04.002. PMC 4433760. PMID 25931378.
- ↑ Doi T, Asada A, Takeda A, Tagami T, Katagi M, Matsuta S, et al. (January 2016). "Identification and characterization of α-PVT, α-PBT, and their bromothienyl analogs found in illicit drug products". Forensic Toxicology. 34 (1): 76–93. doi:10.1007/s11419-015-0288-3. S2CID 24528511.
- ↑ Uchiyama N, Matsuda S, Kawamura M, Kikura-Hanajiri R, Goda Y (July 2013). "Two new-type cannabimimetic quinolinyl carboxylates, QUPIC and QUCHIC, two new cannabimimetic carboxamide derivatives, ADB-FUBINACA and ADBICA, and five synthetic cannabinoids detected with a thiophene derivative α-PVT and an opioid receptor agonist AH-7921 identified in illegal products". Forensic Toxicology. 31 (2): 223–240. doi:10.1007/s11419-013-0182-9. S2CID 1279637.
- ↑ Takayama T, Suzuki M, Todoroki K, Inoue K, Min JZ, Kikura-Hanajiri R, et al. (June 2014). "UPLC/ESI-MS/MS-based determination of metabolism of several new illicit drugs, ADB-FUBINACA, AB-FUBINACA, AB-PINACA, QUPIC, 5F-QUPIC and α-PVT, by human liver microsome". Biomedical Chromatography. 28 (6): 831–838. doi:10.1002/bmc.3155. PMID 24861751.
- ↑ Swortwood MJ, Carlier J, Ellefsen KN, Wohlfarth A, Diao X, Concheiro-Guisan M, et al. (January 2016). "In vitro, in vivo and in silico metabolic profiling of α-pyrrolidinopentiothiophenone, a novel thiophene stimulant". Bioanalysis. 8 (1): 65–82. doi:10.4155/bio.15.237. PMC 5493968. PMID 26648097.
- ↑ Wojcieszak J, Andrzejczak D, Woldan-Tambor A, Zawilska JB (August 2016). "Cytotoxic Activity of Pyrovalerone Derivatives, an Emerging Group of Psychostimulant Designer Cathinones". Neurotoxicity Research. 30 (2): 239–250. doi:10.1007/s12640-016-9640-6. PMID 27295059. S2CID 11202107.
- ↑ "Cannabinoider föreslås bli klassade som hälsofarlig vara" (in Swedish). Retrieved 29 June 2015.
- ↑ "关于印发《非药用类麻醉药品和精神药品列管办法》的通知" (in Chinese). China Food and Drug Administration. 27 September 2015. Archived from the original on 1 October 2015. Retrieved 1 October 2015.
- ↑ "Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien" (in German). Der Bundesrat.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.