 | |
 | |
| Names | |
|---|---|
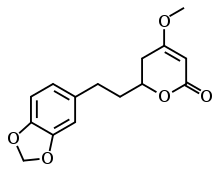
| IUPAC name
(2S)-2-[2-(1,3-benzodioxol-5-yl)ethyl]-4-methoxy-2,3-dihydropyran-6-one | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| MeSH | C107882 |
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C15H16O5 | |
| Molar mass | 276.28 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Dihydromethysticin is one of the six major kavalactones found in the kava plant.[1]
Pharmacology
Dihydromethysticin has marked activity on the induction of CYP3A23, as does the related chemical desmethoxyyangonin.[2]
Both dihydromethysticin and methysticin induce the hepatic enzyme CYP1A1, which increases the amount of the very highly carcinogenic benzo[a]pyrene-7,8-dihydrodiol-9,10-epoxide in the body (via the metabolism of benzo[a]pyrene) and may be responsible for some of the toxic effects associated with kava consumption.
pyrene_metabolism.svg.png.webp)
Metabolism of benzo[a]pyrene yielding the carcinogenic benzo[a]pyren-7,8-dihydrodiol-9,10-epoxide.
In vitro, dihydromethysticin possesses analgesic, anticonvulsant, and anxiolytic effects.[3] It has been found to act as a GABAA receptor positive allosteric modulator and as an reversible inhibitor of monoamine oxidase B.[4][5]
References
- ↑ Malani, Joji (2002-12-03). "Evaluation of the effects of Kava on the Liver" (PDF). Fiji School of Medicine. Archived from the original (PDF) on 2009-03-20. Retrieved 2009-09-04.
- ↑ Ma, Yuzhong; Karuna Sachdeva; Jirong Liu1; Michael Ford; Dongfang Yang; Ikhlas Khan; Clinton Chichester; Bingfang Yan (November 2004). "Desmethoxyyangonin and dihydromethysticin are two major pharmacological kavalactones with marked activity on the induction of CYP3A23". Drug Metabolism and Disposition. 32 (11): 1317–1324. doi:10.1124/dmd.104.000786. PMID 15282211. S2CID 43840844.
{{cite journal}}: CS1 maint: numeric names: authors list (link) - ↑ Walden J, von Wegerer J, Winter U, Berger M, Grunze H (May 1997). "Effects of kawain and dihydromethysticin on field potential changes in the hippocampus". Progress in Neuro-Psychopharmacology and Biological Psychiatry. 21 (4): 697–706. doi:10.1016/s0278-5846(97)00042-0. PMID 9194150. S2CID 34014477.
- ↑ Sarris, Jerome; Laporte, Emma; Schweitzer, Isaac (2011). "Kava: A Comprehensive Review of Efficacy, Safety, and Psychopharmacology". Australian & New Zealand Journal of Psychiatry. 45 (1): 27–35. doi:10.3109/00048674.2010.522554. PMID 21073405. S2CID 42935399.
- ↑ Singh YN, Singh NN (2002). "Therapeutic potential of kava in the treatment of anxiety disorders". CNS Drugs. 16 (11): 731–43. doi:10.2165/00023210-200216110-00002. PMID 12383029. S2CID 34322458.
External links
 Media related to Dihydromethysticin at Wikimedia Commons
Media related to Dihydromethysticin at Wikimedia Commons
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.