 | |
| Names | |
|---|---|
| Preferred IUPAC name
Dimethyl phosphonate | |
| Other names
Phosphonic acid, dimethyl ester | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.011.622 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C2H7O3P | |
| Molar mass | 110.049 g·mol−1 |
| Appearance | colorless liquid |
| Density | 1.20 g/cm3 |
| Boiling point | 72–73 °C (162–163 °F; 345–346 K) 25 Torr |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
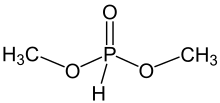
Dimethylphosphite is an organophosphorus compound with the formula (CH3O)2P(O)H, known as dimethyl hydrogen phosphite (DMHP). Dimethylphosphite, is a minor tautomer of the phosphorus(V) derivative. It is a reagent for generating other organophosphorus compounds, exploiting the high reactivity of the P-H bond. The molecule is tetrahedral. It is a colorless liquid. The compounds can be prepared by methanolysis of phosphorus trichloride or by heating diethylphosphite in methanol.[1]
Although studies have not been reported for this compound, the closely related diethylphosphite exists predominantly as the phosphorus(V) tautomer.[2]
This tautomeric nature of DMHP made it desirable as a precursor to the G-series compounds, and it was the most successful among all other phosphonate precursors.[3] The now obsolete process, which used it as a precursor, was called the DMHP process, investigated by Otto Ambros' team and implemented to scale sarin production.[4]
References
- ↑ Balint, Erika; Tajti, Adam; Drahos, Laszlo; Ilia, Gheorge; Keglevich, Gyorgy (2013). "Alcoholysis of Dialkyl Phosphites Under Microwave Conditions". Current Organic Chemistry. 17 (5): 555–562. doi:10.2174/1385272811317050010.
- ↑ Guthrie, J. Peter (1979). "Tautomerization Equilibria for Phosphorous Acid and its Ethyl Esters, Free Energies of Formation of Phosphorous and Phosphonic Acids and their Ethyl Esters, and p Ka Values for Ionization of the P—H Bond in Phosphonic Acid and Phosphonic Esters". Canadian Journal of Chemistry. 57 (2): 236–239. doi:10.1139/v79-039.
- ↑ Sipri - Chemical Weapons: Destruction and Conversion. p 57-62
- ↑ Mark A. Prelas, Dabir S. Viswanath,. Science and Technology of Terrorism and Counterterrorism, Second lllllEdition. p 341-342.