| |||
| Legal status | |||
|---|---|---|---|
| Legal status | |||
| Identifiers | |||
| |||
| CAS Number | |||
| PubChem CID | |||
| ChemSpider |
| ||
| UNII | |||
| ChEMBL | |||
| Chemical and physical data | |||
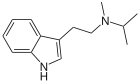
| Formula | C14H20N2 | ||
| Molar mass | 216.328 g·mol−1 | ||
| 3D model (JSmol) | |||
| |||
| |||
| (verify) | |||
N-methyl-N-isopropyltryptamine (MiPT) is a psychedelic tryptamine, closely related to DMT, DiPT and miprocin. It was first synthesized by David Repke in 1984[1] and was subsequently evaluated and described in Alexander Shulgin's 1997 book TiHKAL.
Chemistry
MiPT is the N-isopropyl analog of DMT and the N-methyl analog of DiPT.
MiPT base, unlike many other tryptamines in their freebase form, does not decompose rapidly in the presence of light or oxygen.
In August 2019, Chadeayne et al. solved the crystal structure of fumarate salt of MiPT.[2]
Dosage
Moderate effects have been reported at 10-25 mg ingested orally, with effects lasting 4–8 hours. One of the test subjects in TiHKAL reported moderate effects at 20 mg ingested intranasally.[3]
Effects
In TiHKAL,[4] the subjective experience is reported to be biased towards mental (psychedelic/entheogenic) effects, with mild perceptual (sensory/hallucinogenic) alterations relative to other tryptamines.[3] Subjects reported enhancement of the visual field (brightened and modulated color perception) but a lack of visual distortion typical of tryptamines such as psilocin. Enhancement of auditory perception was also noted. Documented physical effects include stimulation, dry mouth, and muscle tension.
Legality
In the United States, MiPT is unscheduled but purchase, sale, or possession for human consumption could be prosecuted under the Federal Analogue Act.[5] MiPT is specifically mentioned in the DEA Orange Book.[6]
Sweden's public health agency suggested classifying MiPT as a hazardous substance, on May 15, 2019.[7]
See also
References
- ↑ Glennon RA, Jacyno JM, Young R, McKenney JD, Nelson D (January 1984). "Synthesis and evaluation of a novel series of N,N-dimethylisotryptamines". Journal of Medicinal Chemistry. 27 (1): 41–45. doi:10.1021/jm00367a008. PMID 6581313.
- ↑ Chadeayne AR, Pham DN, Golen JA, Manke DR (September 2019). "The fumarate salts of the N-isopropyl-N-methyl derivatives of DMT and psilocin". Acta Crystallographica. Section E, Crystallographic Communications. 75 (Pt 9): 1316–1320. doi:10.1107/S2056989019011253. PMC 6727059. PMID 31523457.
- 1 2 Shulgin A, Shulgin A (September 1997). "#47 MIPT". Isomer Design. Transform Press. Retrieved 28 November 2023.
- ↑ Shulgin, Alexander; Shulgin, Ann (September 1997). TiHKAL: The Continuation. Berkeley, California: Transform Press. ISBN 0-9630096-9-9. OCLC 38503252.
- ↑ "21 U.S. Code § 841 - Prohibited acts A", LII / Legal Information Institute, retrieved 2016-08-02
- ↑ "Orange Book - List of Controlled Substances and Regulated Chemicals" (PDF). Drug Enforcement Administration. Archived (PDF) from the original on 6 March 2023.
- ↑ "Folkhälsomyndigheten föreslår att 20 ämnen klassas som narkotika eller hälsofarlig vara" (in Swedish). Folkhälsomyndigheten. 15 May 2019.
External links
| 5-HT1 |
| ||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT2 |
| ||||||||||||||||||||||||||||||||||||||
| 5-HT3–7 |
| ||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||
|