 | |
| Clinical data | |
|---|---|
| Other names | DMS; (Ε)-α,α'-Dimethyl-4,4'-stilbenediol |
| Drug class | Nonsteroidal estrogen |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
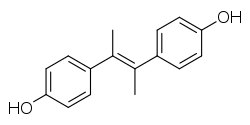
| Formula | C16H16O2 |
| Molar mass | 240.302 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Dimethylstilbestrol (DMS) is a nonsteroidal estrogen of the stilbestrol group related to diethylstilbestrol which was never marketed.[1][2][3][4][5]: 213 It is a so-called "weak", "impeded", or "short-acting" estrogen similarly to estriol and meso-butoestrol.[6][7][8][9] The affinity of DMS for the ER was reported as about 10% of that of estradiol.[10] For comparison, diethylstilbestrol had 140% of the affinity of estradiol for the ER.[10]
The endometrial proliferation dose of DMS in women is 20 mg.[5]: 212–213 A single 12 mg intramuscular injection of DMS has a duration of approximately 12 days in humans.[5]
References
- ↑ Pincus G (3 September 2013). "Blastocyst Development and Implantation". The Control of Fertility. Elsevier. pp. 126–. ISBN 978-1-4832-7088-3.
- ↑ Emmens CW, Martin L (5 December 2016). "Anti-Estrogens". In Dorfman RI (ed.). Steroidal Activity in Experimental Animals and Man. Elsevier Science. pp. 83–. ISBN 978-1-4832-7299-3.
- ↑ Hilf R, Wittliff JL (27 November 2013). "Mechanims of Action of Estrogens". In Sartorelli AC, Johns DG (eds.). Antineoplastic and Immunosuppressive Agents. Springer Science & Business Media. pp. 110–. ISBN 978-3-642-65806-8.
- ↑ Horwitz KB, McGuire WL (14 December 2013). "Antiestrogens: Mechanism of Action and Effects in Breast Cancer". In McGuire W (ed.). Experimental Biology. Springer Science & Business Media. pp. 169–. ISBN 978-1-4757-4673-0.
- 1 2 3 Knörr K, Beller FK, Lauritzen C (17 April 2013). "Prinzipien der Hormonbehandlung: Die wichtigsten hormonalen Behandlungsmethoden". Lehrbuch der Gynäkologie. Springer-Verlag. ISBN 978-3-662-00942-0.
- ↑ Katzenellenbogen BS, Iwamoto HS, Heiman DF, Lan NC, Katzenellenbogen JA (1978). "Stilbestrols and stilbestrol derivatives: estrogenic potency and temporal relationships between estrogen receptor binding and uterine growth". Molecular and Cellular Endocrinology. 10 (1): 103–113. doi:10.1016/0303-7207(78)90063-1. PMID 564791. S2CID 45882988.
- ↑ Martin L (January 1969). "Dimethylstilbestrol and 16-oxo-estradiol: anti-estrogens or estrogens?". Steroids. 13 (1): 1–10. doi:10.1016/s0039-128x(69)80055-3. PMID 5764482.
- ↑ Emmens CW, Cox RI, Martin L (July 1959). "Oestrogen inhibitors of the stilboestrol series". The Journal of Endocrinology. 18 (4): 372–380. doi:10.1677/joe.0.0180372. PMID 13820198.
- ↑ Emmens CW, Cox RI (September 1958). "Dimethylstilboestrol as an oestrogen inhibitor". The Journal of Endocrinology. 17 (3): 265–271. doi:10.1677/joe.0.0170265. PMID 13587831.
- 1 2 Jordan VC, Lieberman ME (September 1984). "Estrogen-stimulated prolactin synthesis in vitro. Classification of agonist, partial agonist, and antagonist actions based on structure". Molecular Pharmacology. 26 (2): 279–285. CiteSeerX 10.1.1.1064.9508. PMID 6541293.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.